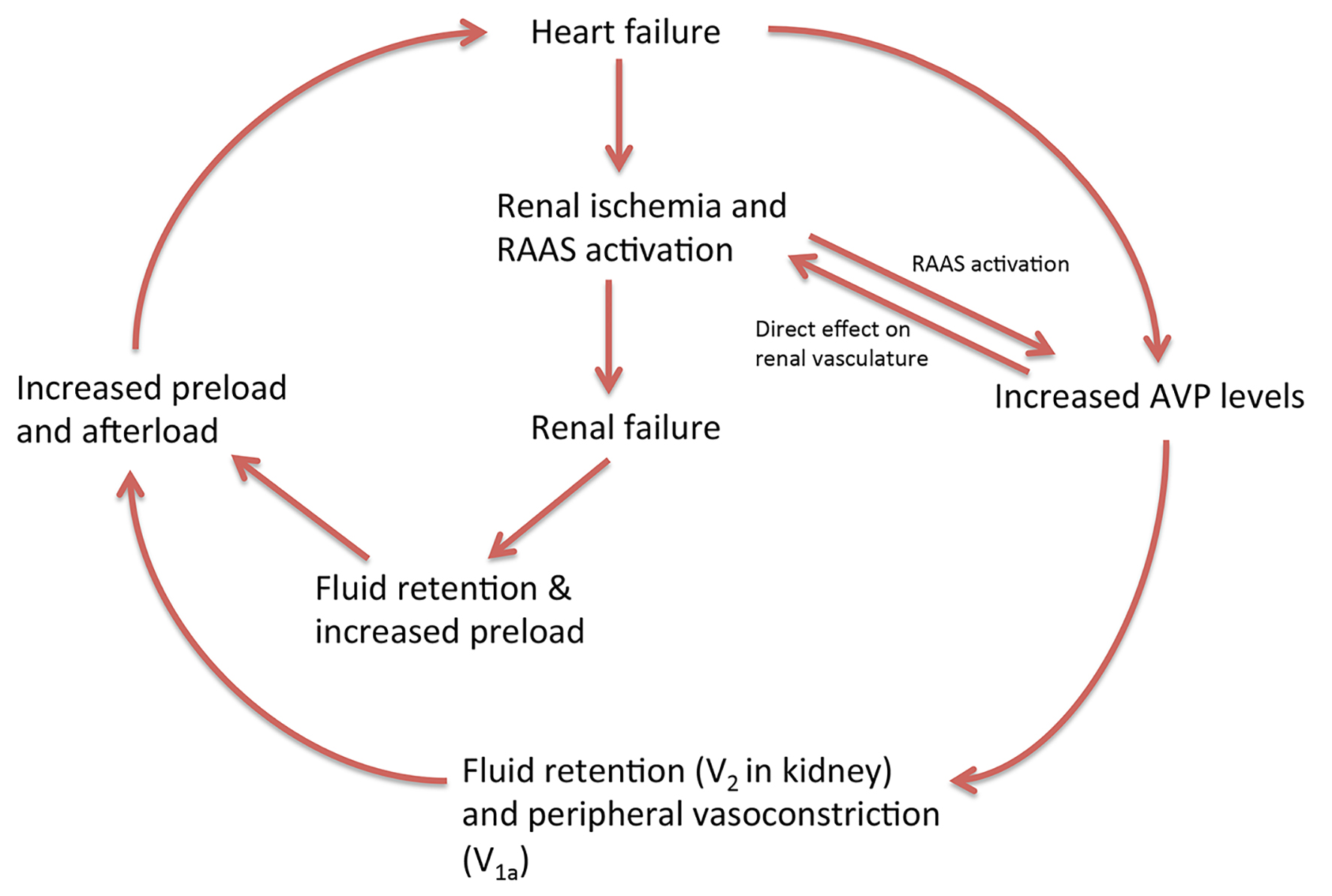
Figure 1. Vicious cycle involving heart, kidney and arginine vasopressin. RAAS: renin-angiotensin-aldosterone system; AVP: arginine vasopressin.
| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Review
Volume 8, Number 3, June 2017, pages 87-95
Cardiorenal Syndrome: Role of Arginine Vasopressin and Vaptans in Heart Failure
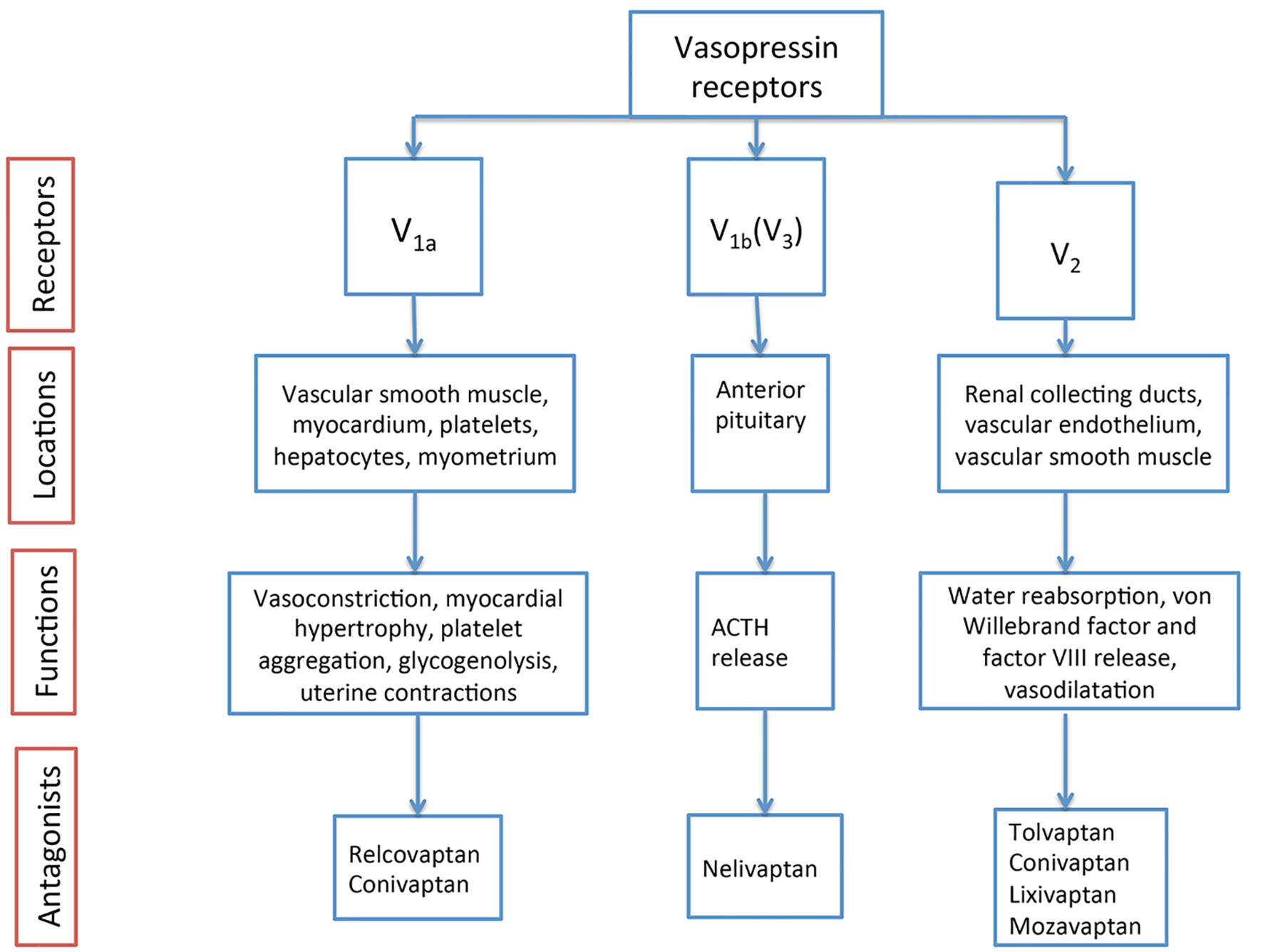
Figures

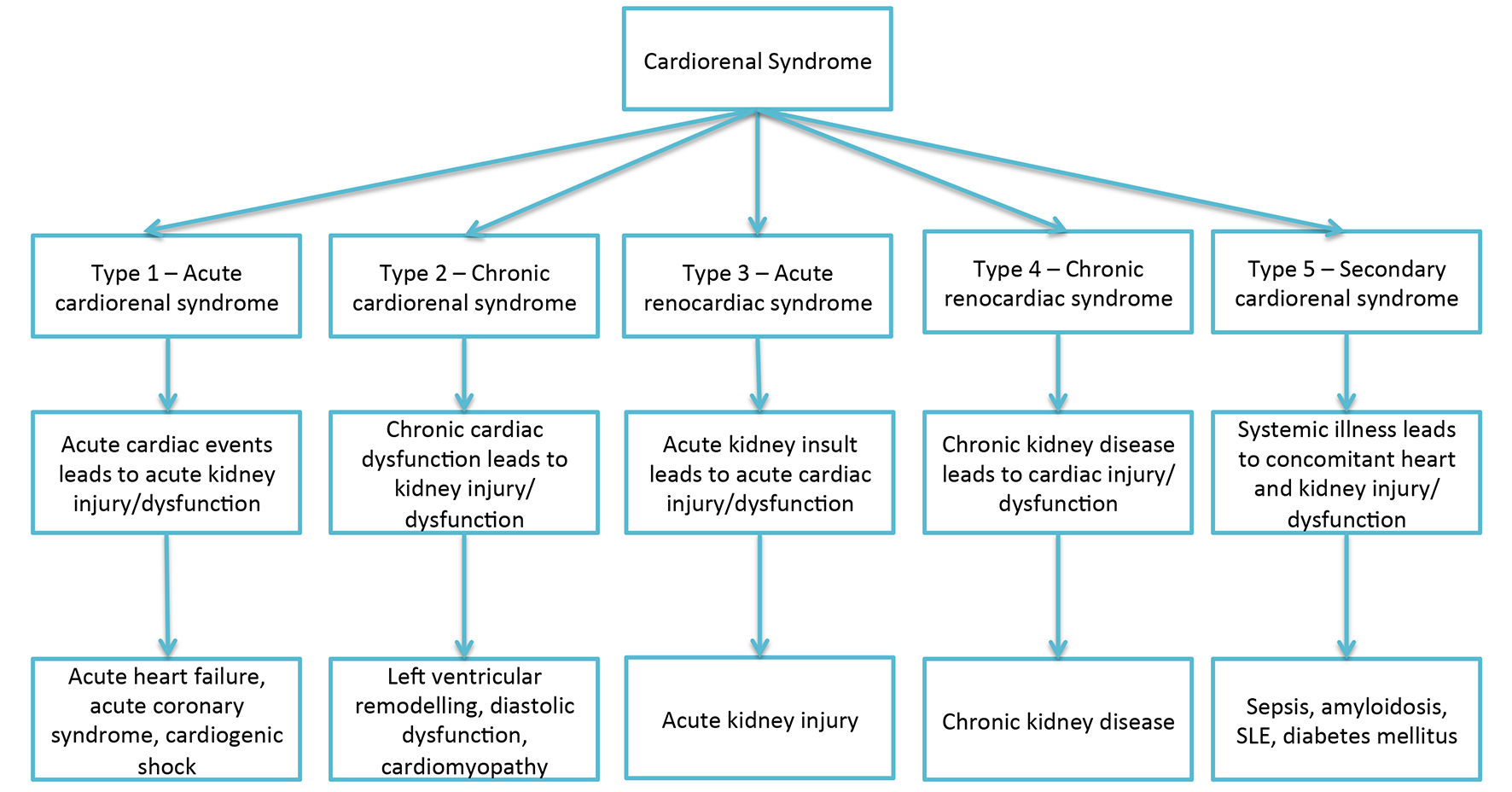
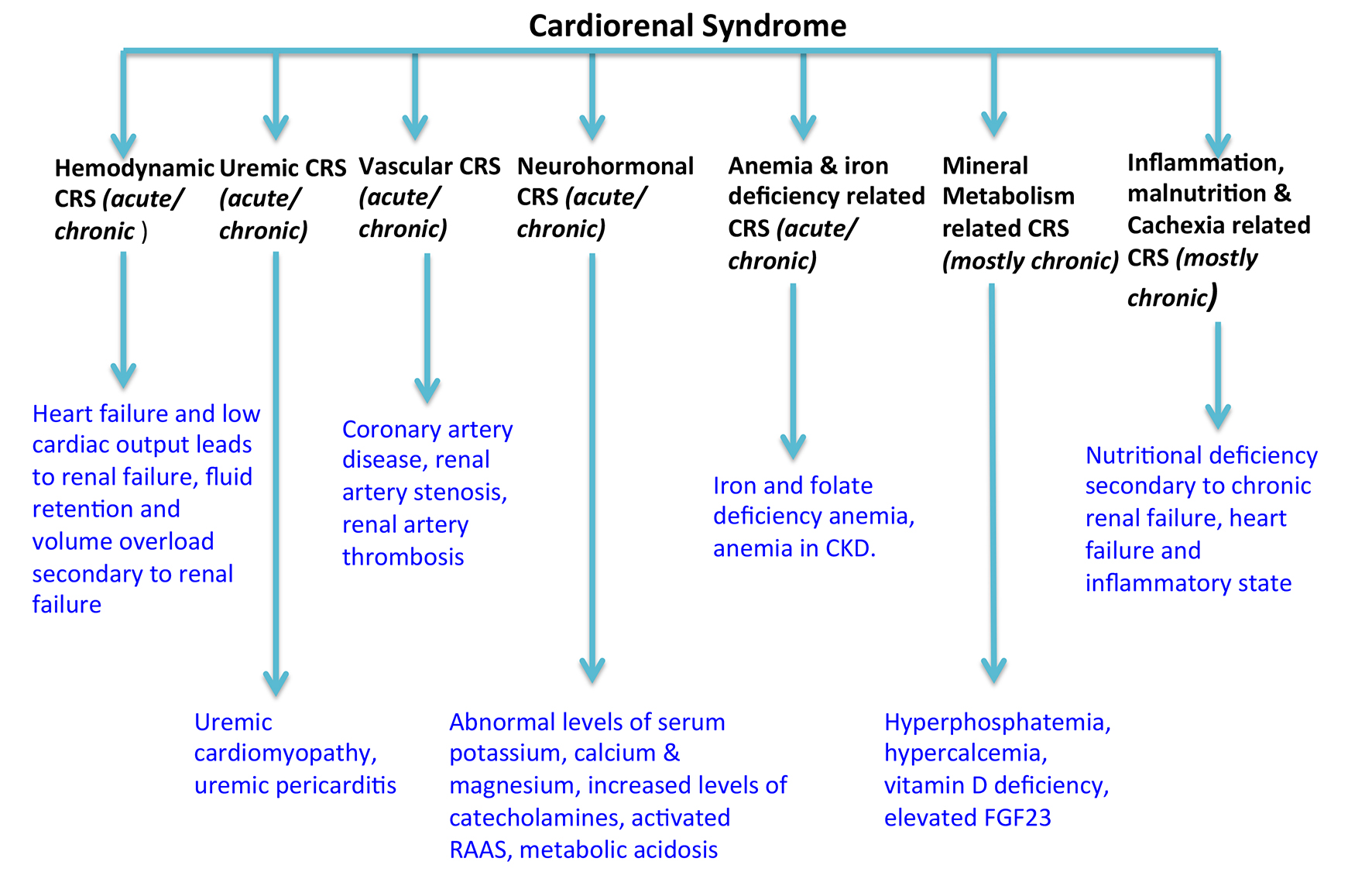
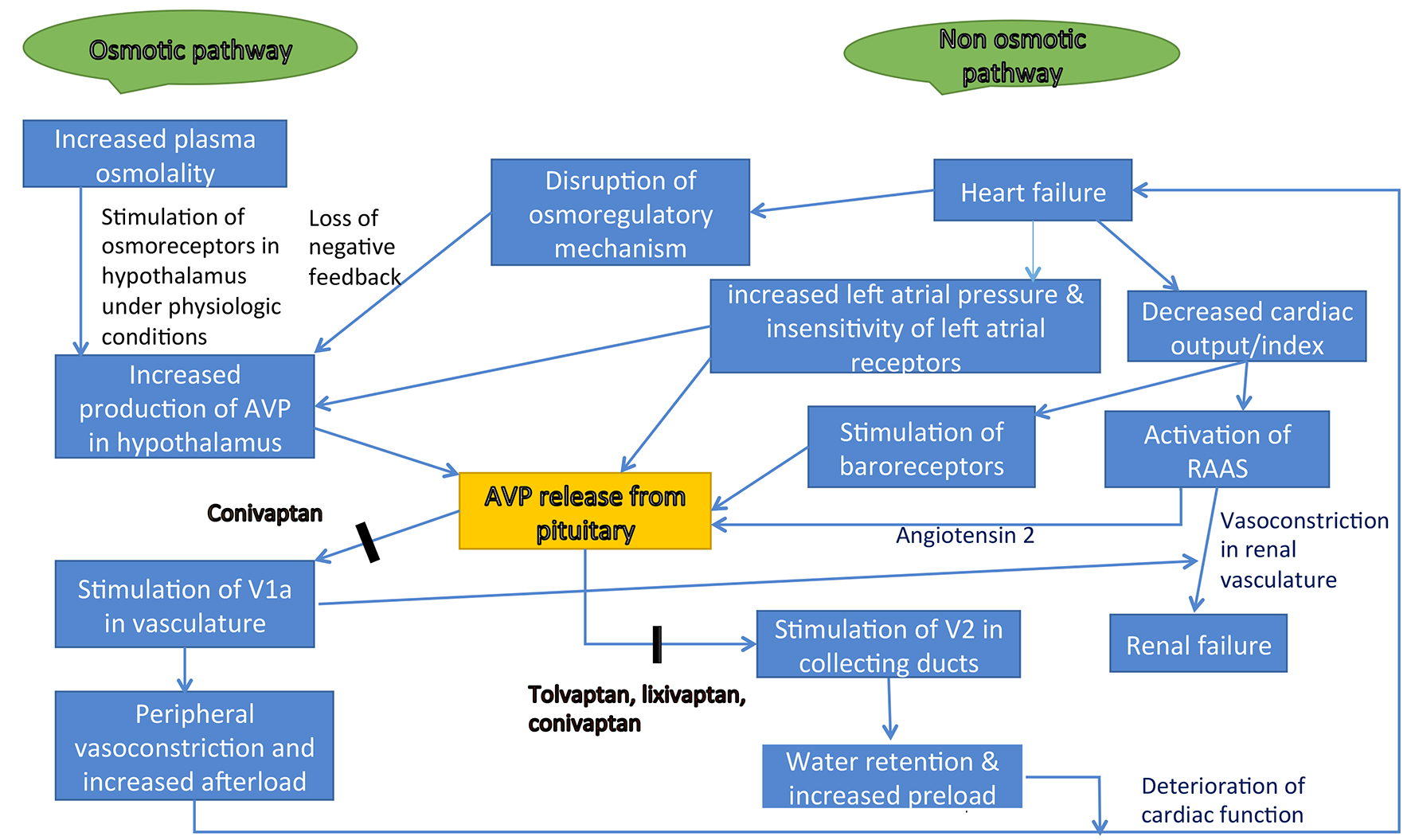
Table
| Study | Drug | No. of subjects | Clinical characteristics | Outcome |
|---|---|---|---|---|
| Udelson et al (2001) [26] Randomized double-blind trial | Conivaptan | Total 142 patients (38 received placebo, 37 received 10 mg, 32 received 20 mg, and 35 received 40 mg) | Symptomatic heart failure, NYHA class III and IV. Baseline serum creatinine 1.1 - 1.3 mg/dL. Patients with serum creatinine > 2.5 mg/dL or creatinine clearance < 30 mL/min were excluded from the study. | Dose dependent diuresis with no change in BP, HR or serum electrolytes. Reduction in PCWP and right atrial pressure due to direct myocardial V1a receptor blockade and diuresis secondary to V2 receptor. |
| Palmer et al (2016) [30] Open label multicenter study | Conivaptan | Total 251 patients (37 received 20 mg/day and 214 received 40 mg/day) | Euvolemic or hypervolemic hyponatremia patients (majority are heart failure and SIADH, some are unknown cause). Patients with eGFR < 20 mL/min were excluded from the study. | Both the dose regimens are equally efficacious and well tolerated with infusion site reaction as the common side effect. |
| Gheorghiade et al (2003) [31] Double-blind placebo controlled trial | Tolvaptan | Total 254 patients (63 received placebo, 64 received 30 mg/day, 64 received 45 mg/day and 63 received 60 mg/day) | Congestive heart failure patients, NYHA class I-IV. Patients with serum creatinine > 3 mg/dL and BUN > 60 mg/dL were excluded from the study. | Increased urine output, restoration of serum Na+ levels without any change in BP, HR, serum K+ or renal function when added to standard therapy |
| Schrier et al (2006) [29] Multicenter, randomized, double-blind, placebo-controlled trials (SALT 1 and SALT 2) | Tolvaptan | Total 448 patients (225 received 15 mg/day and later increased to 30 mg/day and then to 60 mg/day depending on serum Na+ levels) | Euvolemic or hypervolemic hyponatremia patients (heart failure, cirrhosis, SIADH). Patients with serum creatinine > 3.5 mg/dL were excluded from the study. | Effective dose-dependent correction of hyponatremia when added to standard therapy |
| Konstam et al (2007) [32] The EVEREST outcome trial. Randomized, double-blind, placebo-controlled trial | Tolvaptan | Total 4,133 patients (2,072 received 30 mg/day and 2,061 received placebo) | Congestive heart failure patients, NYHA class III and IV. Patients with serum creatinine > 3.5 mg/dL were excluded from the study. | No mortality or morbidity benefits when added to standard therapy. Short-term benefits observed were weight loss, improvement in symptoms, and restoration of serum Na+ levels with no change in renal functions. |
| Udelson et al (2007) [33] Multicenter, randomized, double-blind, placebo-controlled trial | Tolvaptan | Total 240 patients (120 received 30 mg/day and 120 received placebo) | Heart failure patients, NYHA II and III with EF < 30%. Patients with serum creatinine > 3 mg/dL and BUN > 60 mg/dL were excluded from the study. | Long-term use for a period of one year is safe and well tolerated with better mortality and morbidity rate. No change in renal function or serum electrolytes observed. No effect on ventricular remodeling |
| Nakada et al (2015) [34] Retrospective study | Tolvaptan | Total 206 patients (26 conventional diuretic resistant patients received tolvaptan and 180 received conventional diuretic therapy) | Acute heart failure patients | Dilated left atrium and inferior vena cava, and severe tricuspid incompetence are frequently associated findings in acute heart failure patients who need tolvaptan therapy. |
| Tanaka et al (2015) [35] Prospective study | Tolvaptan | Total 20 patients | Volume overload due to CKD. In addition, few patients with heart and liver failure. | Effective diuresis without deterioration in renal functions |
| Shanmugam et al (2016) [36] Randomized, double-blind, placebo-controlled trial | Tolvaptan for 5 days | Total 51 patients (26 received placebo and 25 received 15 mg/day) | Acute decompensated heart failure with hyponatremia. Patients with serum creatinine > 3 mg/dL were excluded from the study. | Increased urine output and restoration of serum Na+ levels when added to standard therapy |