| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Original Article
Volume 11, Number 3, June 2020, pages 179-184
Is the Time of Atrial Fibrillation Recurrence After Duty-Cycled Radiofrequency Ablation Affected by the Pattern of Pulmonary Vein Reconnections?
Marcus Wieczoreka, b, c, Kiarash Sassanib, Reinhard Hoeltgenb
aSchool of Medicine, Witten/Herdecke University, Witten, Germany
bDepartment of Cardiology and Electrophysiology, St. Agnes-Hospital Bocholt, Barloer Weg 125, Bocholt 46397, Germany
cCorresponding Author: Marcus Wieczorek, St. Agnes-Hospital Bocholt, Barloer Weg 125, Bocholt 46397, Germany
Manuscript submitted March 10, 2020, accepted March 31, 2020
Short title: Time of AF Recurrence and PV Reconnection
doi: https://doi.org/10.14740/cr1050
| Abstract | ▴Top |
Background: Pulmonary vein isolation (PVI) with multielectrode duty-cycled radiofrequency (PVAC) has been shown to be effective in the treatment of atrial fibrillation (AF). We describe pulmonary vein (PV) reconnection at repeat ablation in patients with AF recurrence after PVAC PVI and analyze the correlation between the time of AF recurrence and the observed PV reconnection patterns.
Methods: Eighty-five patients undergoing a redo PVI for recurrent AF 9.2 ± 3.8 months after an initial PVAC PVI procedure was retrospectively enrolled.
Results: A total of 93% had PV reconnections with a mean of 2.97 ± 1.2 reconnected PVs/patients and 75% of formerly isolated PVs were found reconnected. The highest reconnection rates (94%) were observed for left common trunks (CTs). A total of 33% patients had three and four reconnected PVs, respectively, while 7% were without PV reconnection. There was a moderate but significant negative correlation between the time of AF recurrences and the extending of PV reconnections at redo PVI for patients with proven PV reconnection (r = -0.32, P = 0.005), whereas five out of six patients without PV reconnection had recurrences within the first 9 months after PVI.
Conclusions: At redo ablation most patients with recurrence of AF after PVAC PVI had PV reconnection(s). Patients with PV reconnection(s) showed a moderate negative correlation between the number of reconnected PVs and the time of AF recurrence with more extensive PV reconnections resulting in earlier PV recurrences after the blanking period. Patients without PV reconnection experienced early AF recurrences, indicating non-PV triggers contribute to AF recurrences in these patients.
Keywords: Atrial fibrillation; Recurrence; Pulmonary vein isolation; Duty-cycled radiofrequency; Reconnection
| Introduction | ▴Top |
Pulmonary vein isolation (PVI) has proven efficacy in the treatment of atrial fibrillation (AF) [1]. Recovery of pulmonary vein (PV) to left atrium (LA) conduction is a frequent electrophysiological finding in patients with recurrent AF after PVI [2, 3]. Repeat PVI can result in a significant improvement in long-term ablation success in these patients [4].
However, little is known about the relationship between the time of AF recurrence and the PV reconnection patterns found at repeat electrophysiological study in patients with AF recurrences after initially successful PVI with multielectrode duty-cycled radiofrequency (PVAC). PVAC has shown to be safe and effective in PVI procedures [5-7]. We hypothesized that more extensive PV reconnections are correlated with an earlier onset of AF recurrence after PVAC PVI.
| Materials and Methods | ▴Top |
Patients
This study was approved by the local ethics committee/review board of Witten/Herdecke University, Germany. All patients in our institution with paroxysmal AF undergoing a second electrophysiology study for AF recurrences after completing a blanking period of 3 months, with a first PVI performed between May 2013 and June 2018, were prospectively evaluated. Energy source for first PVI in our institution was multielectrode duty-cycled radiofrequency (PVAC or PVAC GOLD, Medtronic, CA, USA) in 85 patients. Patients with a need for touch-up ablation during the first procedure were excluded from this study. Only patients completing regular follow-up visits in our institution until recurrence of AF were recruited for data analysis.
Duty-cycled radiofrequency PVI
Our general ablation protocol for PVI using the PVAC has been described in detail elsewhere [8]. In brief, PVAC and PVAC GOLD are steerable, 9-F, over-the-wire circular mapping and ablation catheters used in conjunction with the multichannel radiofrequency (RF) generator (GENius, Medtronic, CA, USA). The system is capable of synchronous application of duty-cycled phased unipolar and bipolar RF energy over all 10 (PVAC) or nine (PVAC GOLD) electrodes, where bipolar refers to adjacent electrodes. After informed consent, PVAC was advanced into the LA using a long steerable 9Fr sheath (Greatbatch Medical™, MN, USA) and directed over a guidewire placed in each of the PVs towards the PV antrum. Bipolar electrograms of PV potentials were recorded over all electrodes. After deep analgosedation, ablation was always begun in a 2:1 bipolar/unipolar mode. It was at the operator’s decision to switch to a 1:1 mode, if PVI could not be reached. Several overlapping RF applications, 60 s each, were made while repositioning the PVAC, until all PV potentials were eliminated. PVAC was also used for validation of PV bidirectional conduction block after a waiting period of 30 min.
Definition and ablation strategy of left common trunk (CT)
CT was defined as the presence of bifurcated PVs entering the LA contour together and at a distance between the virtual border of the LA and the bifurcation of both PVs > 5 mm.
As for CT isolation with PVAC, to avoid PV stenosis of the targeted CTs, PVAC was always directed to the antral aspect of the CTs. Multiple PVAC rotations around the CT ostium followed by energy applications were generally performed, until CT isolation was achieved.
Repeat ablation procedure
All procedures were performed after informed consent and started under conscious sedation. A hexapolar electrode catheter was placed in the coronary sinus (CS) for pacing and bipolar recordings. After transseptal puncture, a long steerable sheath (FlexCath or Greatbatch Medical) was inserted into the LA to delineate all PVs angiographically. All PVs were then assessed for PV-LA reconnection. PV reconnection was determined at the onset of the procedure using a 20-mm inner lumen mapping catheter Achieve (Medtronic, Minneapolis, MN, USA). The catheter was placed in each PV with evaluation for PV potentials. Electric isolation of PVs was confirmed by entrance and exit block for each PV, assessed by Achieve, positioned at the PV antrum and inside the PV, as necessary.
All PVs/CTs with PV-LA reconnection were targeted for re-isolation with second generation cryoballoon (CB) and/or open-irrigated RF-energy point by point, depending on the operator’s preference.
Follow-up strategy
All patients were routinely seen every 3 months after the initial ablation procedure in an outpatient clinic or earlier in case of sustained palpitations. Antiarrhythmic drug (AAD) therapy was used in patients with early and recurrent episodes of symptomatic atrial fibrillation and discontinued again after a blanking period of 3 months at least 7 days before the first follow-up evaluation. Therapy with amiodarone, if transiently prescribed after the first AF recurrence, was discontinued at the time of re-ablation. Five-day Holter monitor recordings were performed off AAD therapy at each follow-up interval, starting 3 months after the initial procedure. Patients were further requested to visit our hospital in the event of ongoing palpitations. Freedom from AF was defined as the absence of atrial fibrillation lasting > 30 s.
Statistical analysis
All continuous variables are reported as mean ± standard deviation (SD) and were compared by Student’s t-test, Mann-Whitney U test or Wilcoxon test. Categorical variables were compared by Chi-square or Fisher’s exact method, as appropriate. A two-tailed P value < 0.05 was considered to be statistically significant. Bivariate correlation was calculated using Pearson’s correlation coefficient. A linear regression model was used for significance analysis. All statistical analysis was performed using GraphPad Prism software, version 8.
| Results | ▴Top |
Study population
As a result of the exclusion criteria, a total of 85 patients were finally included in this study with a mean age of 58 ± 17 years, male dominance (66%), moderate LA enlargement (43 ± 4 mm) and mean CHA2DS2-VASc-score 2.2. A total of 49% of the patients were diagnosed with hypertension (Table 1).
 Click to view | Table 1. Clinical Characteristics of the Enrolled Patients (n = 85) |
Reconnection patterns at repeat ablation
Average time until recurrence of AF after ablation was 8.1 ± 3.4 months. After a mean 9.2 ± 3.8 months, a redo ablation procedure was performed in all 85 patients. Electrophysiological characteristics at the time of re-ablation are presented in Table 2: 93% of the patients had PV reconnection(s) and 237 out of 315 PVs/CTs (75%) were found reconnected resulting in a mean of 2.79 ± 1.2 reconnected PVs/patient.
 Click to view | Table 2. Electrophysiological Characteristics at Redo Procedure |
A detailed analysis of LA-PV reconnections for each PV is presented in Table 2: the lowest reconnection rate after PVAC PVI was observed in the right inferior pulmonary veins (RIPVs) (58%), while the highest rate of reconnection was detected for CTs (94%).
We observed the following distribution of patients with 0, 1, 2, 3 and 4 reconnected PVs at the time of redo PVI: 28 (33%) patients had four and three reconnected PVs, respectively. Thus, the percentage of patients with ≥ 3 reconnected PVs was 66% and observed in 56 patients, while the percentage of patients with no PV reconnection (n = 6) was low (7%). A minority of patients (n = 5) had only one reconnected PV (6%).
Time course of AF recurrences and PV reconnection patterns
The majority of patients (n = 64, 75%) experienced AF recurrences early between month 4 and 9 follow-up, compared with 21 patients having AF recurrences later (≥ 10 months) after PVI (P < 0.0001). Ten patients were on transient amiodaron therapy during the blanking period: 7/64 patients with early and 3/21 patients with late AF recurrences (P = 0.7030).
The relationship between time of AF recurrence and the number of reconnected PVs at redo PVI is presented in Table 3: five out of six patients without PV reconnection at redo PVI had AF recurrences within the first 9 months. Patients with three or four reconnected PVs were most frequently observed during redo PVI (n = 28 in each group). These patients had the highest risk of AF recurrences 4 - 9 months after PVI: 20/28 patients with three and 21/28 patients with four reconnected PVs, respectively. Patients with two reconnected PVs had a maximum of AF recurrences between 7 and 9 months after PVI (12/18 patients).
 Click to view | Table 3. Time of AF Recurrence and Number of Reconnected PV at Redo Ablation in 85 Patients |
Comparing early (4 - 9 months) with late (10 months or later) AF recurrences, we have the results about the differences presented in Figure 1: no differences between early and late AF recurrences were observed for patients with no PV reconnection and for patients with one reconnected PV. However, patients with more than one reconnected PV experienced AF recurrences significantly earlier with P < 0.001 for patients with two and four reconnected PVs and P < 0.05 for patients with three reconnected PVs.
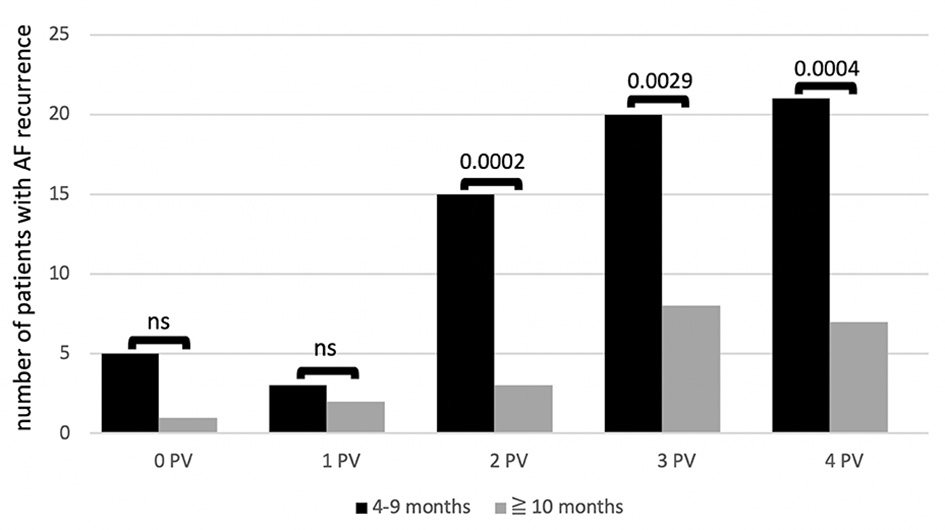 Click for large image | Figure 1. Number of patients with recurrence of AF during follow-up depending on number of reconnected PVs (0 - 4) found at redo PVI at different time intervals: early recurrence (black column) versus late recurrence (grey column). P values above each column. Left common trunk counted as one PV. AF: atrial fibrillation; ns: not significant: PV: pulmonary vein; PVI: pulmonary vein isolation. |
Correlating all AF recurrences of patients with reconnected PVs (n = 79) with the extending of PV reconnection found at redo PVI, we found a moderate but significant negative correlation between extending of reconnections and the time of AF recurrence (r = -0.32, P = 0.0045), which is shown in Figure 2.
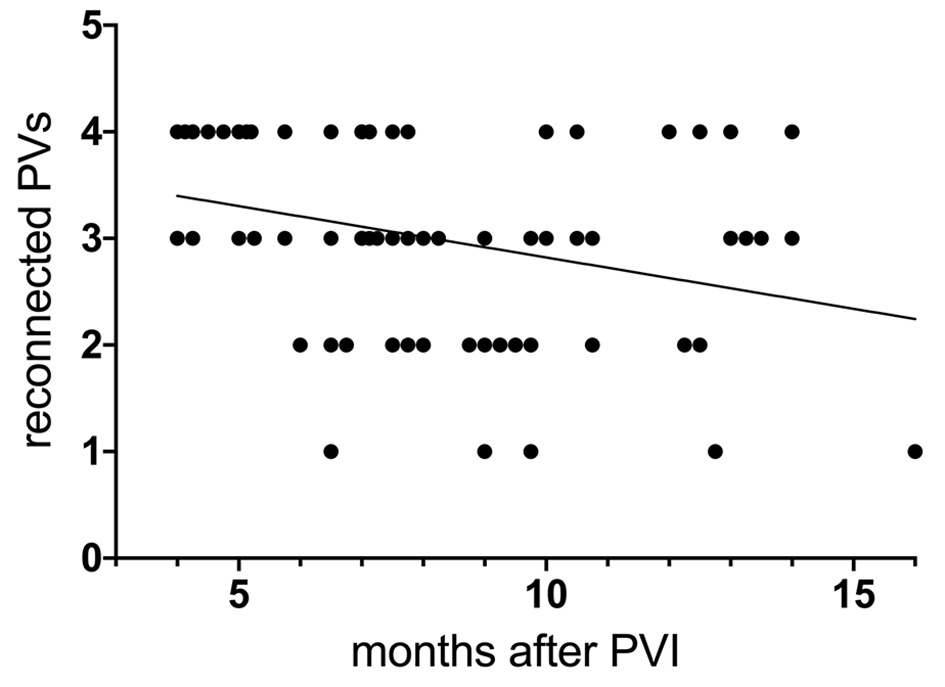 Click for large image | Figure 2. Correlation between time of AF recurrence after PVAC PVI and number of reconnected PVs at redo ablation with corresponding regression line. Left common trunk counted as one PV. PV: pulmonary vein; PVI: pulmonary vein isolation; AF: atrial fibrillation; PVAC: multielectrode duty-cycled radiofrequency. |
| Discussion | ▴Top |
Main findings
We analyzed LA-PV reconnection patterns in 85 patients with recurrent AF undergoing repeat PVI. Initial PVI had been performed by PVAC and PVAC GOLD. After a mean of 9.2 months, a high percentage of patients (93%) had PV reconnection in one or more PVs with 66% of all patients presenting with three or four reconnected PVs. CTs (94%) and left superior pulmonary veins (LSPVs) (85%) were found most frequently reconnected after PVI with PVAC.
The majority of patients without PV reconnection (7%) had early AF recurrences within 9 months. However, AF recurrences in patients with more than one reconnected PV occurred more frequently within the first 9 months after PVI compared with recurrences beyond this interval.
In brief, the extending of PV reconnections found at redo PVI seems to have affected the time of AF recurrence after PVI in the majority of patients, resulting in a moderate but significant negative correlation between time of AF recurrence and extending of PV reconnection for patients with reconnected PV(s).
PV reconnection patterns after PVAC PVI
PV reconnection is the typical finding in patients with recurrent AF after PVI. Different PV reconnection rates (19% after second generation CB and 64% after PVAC) in patients with recurrent AF have been reported in the past for other ablation methods [9, 10]. A total of 75% reconnected PVs and CTs after PVAC PVI in our study indicate an even higher PV reconnection rate at redo PVI compared with 64% notified by Balt et al [10].
In our study PV reconnections after PVAC PVI were most frequently observed in CTs and LSPVs. This finding is consistent with data from CB ablation presented by Shah et al and Glowniak et al [11, 12], while Aryana et al found that CTs most frequently reconnected after CB PVI [9].
A total of 60% of reconnected CTs were observed in our series of patients. This finding is believed to reflect the compromised ability to routinely achieve optimal contact between a fixed circumferential catheter of 25 mm and CTs of greater diameter. As a result, multiple PVAC rotations and energy applications resulting in inhomogeneous overlapping antral lesions are often necessary for CT isolation. Compared with other series of patients undergoing PVI, the percentage of patients with CTs in our study was high (20%), which might have contributed to a higher overall PV reconnection rate.
The majority of patients in our study (66%) had PV reconnections affecting three or four PVs and the average reconnection ratio after PVAC PVI was high with 2.8 PVs/patient indicating that PVAC lesions are not persistent/stable over time in many patients.
Patterns of PV reconnection and time of AF recurrence
It is well known from prior studies that most patients with AF recurrences after PVI have PV reconnections at the time of redo PVI and that PVI re-isolation can improve freedom from AF [4]. On the other hand, there are clinical data indicating that PV reconnection patterns of patients with and without AF recurrence after PVI do not differ significantly [13, 14], suggesting additional mechanisms beyond re-PVI may result in improved clinical outcome, such as antral substrate modification in the course of PV re-isolation and/or elimination of focal antral triggers.
We found a moderate, but significant negative correlation between the extending of PV reconnection in patients with AF recurrences after PVAC PVI undergoing a redo procedure and the time of recurrence of clinical AF for patients with PV reconnection. To the best of our knowledge, this is the first study to associate the time of AF recurrence after PVAC PVI with patterns of PV reconnection. We found that patients with more extensive PV reconnections (> 1 PV) experienced AF recurrences significantly earlier than patients with a singular reconnected PV during follow-up. Further, the vast majority of patients with four reconnected PVs developed AF recurrences within the first 3 months after the blanking period. This phenomenon could be explained as follows. First, an increasing number of reconnected PVs during follow-up increases the probability for reconnection of an arrhythmogenic PV causing AF. Second, with an increasing number of reconnected PVs, the feasibility of triggering (silent) arrhythmogenic foci inside each reconnected PV by other rapidly firing PVs reconnected to LA is enhanced, resulting in a complex interaction of firing and triggering between reconnected PVs. This mechanism of AF initiation/maintenance has been suggested by Kumagai et al, who found that rapid focal activation originating from one PV can trigger a focus in another PV [15]. They postulated that the mechanism of rapid focal activation may be abnormal automaticity or triggered activity. If the latter, the first rapid focal activation from a PV could trigger the next rapid focal activation in another PV, an interaction which may contribute to the maintenance of AF. Thus, rapid focal activation arising from PVs may be important not only as a trigger of onset, but also in the maintenance of AF.
It is estimated that the majority of patients with a paroxysmal character of AF have triggering ectopy originating in the PVs. Haissaguerre et al first reported the essential role of PVs in the initiation and maintenance of paroxysmal AF [16]. Ectopic activities originating from the PVs were identified in 94% of patients suffering from frequent pre-procedural AF episodes. As a result, isolation of all PVs with proven LA-PV connection is now a generally accepted interventional approach for paroxysmal AF [17]. With this empirical approach, the individual mechanism of AF initiation and/or perpetuation is generally not addressed. This could explain the observation in our study that patients without PV reconnection tend to have early AF recurrences, as the mechanism of AF in these patients may in fact be related to extra PV ectopy. Additional ectopic sources triggering AF were identified outside the PVs. They can be located in the vena cavae, crista terminalis, CS, ligament of Marshall, inter-atrial septum, the appendages and others [18, 19]. Lin et al identified non-PV triggers in 20% of the ectopic beats initiating paroxysmal AF [19].
Study limitations
First, this study represents a single center analysis of patients undergoing a repeat procedure following an index PVI using PVAC. As such, unadjusted confounders may have been present and impacted our findings. Patients with asymptomatic recurrence of atrial arrhythmias or shorter AF episodes may have gone undetected and were therefore excluded from this analysis. There was no control group of patients without AF recurrences after PVI undergoing a second electrophysiological study. Therefore, the pathophysiological impact of our findings with the view to AF recurrences correlated with PV reconnections patterns and time of AF recurrence is limited. As there was no adenosine challenge during PV mapping, concealed reconnection might remain undetected in some cases which could have affected the presented PV reconnection patterns and the resulting correlation with AF recurrences over time [20].
Conclusions
At redo ablation most patients with recurrence of AF after PVAC PVI had PV reconnection(s). For patients with proven PV reconnection at redo PVI there was a moderate, but significant negative correlation between the number of reconnected PVs and the time of AF recurrence with more extensive PV reconnections resulting in earlier PV recurrences after the blanking period. Patients without PV reconnection experienced early AF recurrences, indicating non-PV triggers contributing to AF recurrences in these patients.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Written informed consent was obtained from all patients in this study.
Author Contributions
MW, RH and KS equally performed invasive studies. KS analyzed and interpreted the patient data for PVAC, and RH analyzed follow-up data. MW was the major contributor in writing the manuscript. All authors read and approved the final manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Haissaguerre M, Shah DC, Jais P, Hocini M, Yamane T, Deisenhofer I, Garrigue S, et al. Mapping-guided ablation of pulmonary veins to cure atrial fibrillation. Am J Cardiol. 2000;86(9A):9K-19K.
doi - Ouyang F, Antz M, Ernst S, Hachiya H, Mavrakis H, Deger FT, Schaumann A, et al. Recovered pulmonary vein conduction as a dominant factor for recurrent atrial tachyarrhythmias after complete circular isolation of the pulmonary veins: lessons from double Lasso technique. Circulation. 2005;111(2):127-135.
doi pubmed - Verma A, Kilicaslan F, Pisano E, Marrouche NF, Fanelli R, Brachmann J, Geunther J, et al. Response of atrial fibrillation to pulmonary vein antrum isolation is directly related to resumption and delay of pulmonary vein conduction. Circulation. 2005;112(5):627-635.
doi pubmed - Callans DJ, Gerstenfeld EP, Dixit S, Zado E, Vanderhoff M, Ren JF, Marchlinski FE. Efficacy of repeat pulmonary vein isolation procedures in patients with recurrent atrial fibrillation. J Cardiovasc Electrophysiol. 2004;15(9):1050-1055.
doi pubmed - Wieczorek M, Hoeltgen R, Akin E, Salili AR, Oral H, Morady F. Results of short-term and long-term pulmonary vein isolation for paroxysmal atrial fibrillation using duty-cycled bipolar and unipolar radiofrequency energy. J Cardiovasc Electrophysiol. 2010;21(4):399-405.
doi pubmed - Kozluk E, Balsam P, Peller M, Kiliszek M, Lodzinski P, Piatkowska A, Malkowska S, et al. Efficacy of multi-electrode duty-cycled radiofrequency ablation in patients with paroxysmal and persistent atrial fibrillation. Cardiol J. 2013;20(6):618-625.
doi pubmed - Leitz P, Guner F, Wasmer K, Foraita P, Pott C, Dechering DG, Zellerhoff S, et al. Data on procedural handling and complications of pulmonary vein isolation using the pulmonary vein ablation catheter GOLD(R). Europace. 2016;18(5):696-701.
doi pubmed - Wieczorek M, Hoeltgen R, Brueck M. Does the number of simultaneously activated electrodes during phased RF multielectrode ablation of atrial fibrillation influence the incidence of silent cerebral microembolism? Heart Rhythm. 2013;10(7):953-959.
doi pubmed - Aryana A, Singh SM, Mugnai G, de Asmundis C, Kowalski M, Pujara DK, Cohen AI, et al. Pulmonary vein reconnection following catheter ablation of atrial fibrillation using the second-generation cryoballoon versus open-irrigated radiofrequency: results of a multicenter analysis. J Interv Card Electrophysiol. 2016;47(3):341-348.
doi pubmed - Balt JC, Karadavut S, Mulder AA, Luermans JG, Wijffels MC, Boersma LV. Conduction recovery in patients with recurrent atrial fibrillation after pulmonary vein isolation using multi-electrode duty cycled radiofrequency ablation. J Interv Card Electrophysiol. 2013;37(2):197-204.
doi pubmed - Shah S, Xu W, Adelstein E, Voigt A, Saba S, Jain S. Characterization of pulmonary vein reconnection post Cryoballoon ablation. Indian Pacing Electrophysiol J. 2019;19(4):129-133.
doi pubmed - Glowniak A, Tarkowski A, Fic P, Wojewoda K, Wojcik J, Wysokinski A. Second-generation cryoballoon ablation for recurrent atrial fibrillation after an index procedure with radiofrequency versus cryo: Different pulmonary vein reconnection patterns but similar long-term outcome-Results of a multicenter analysis. J Cardiovasc Electrophysiol. 2019;30(7):1005-1012.
doi pubmed - Nery PB, Belliveau D, Nair GM, Bernick J, Redpath CJ, Szczotka A, Sadek MM, et al. Relationship between pulmonary vein reconnection and atrial fibrillation recurrence: a systematic review and meta-analysis. JACC Clin Electrophysiol. 2016;2(4):474-483.
doi pubmed - Jiang RH, Jiang CY. Pulmonary Vein Reconnection in Patients With and Without Atrial Fibrillation Recurrence After Ablation. JACC Clin Electrophysiol. 2016;2(4):484-486.
doi pubmed - Kumagai K, Yasuda T, Tojo H, Noguchi H, Matsumoto N, Nakashima H, Gondo N, et al. Role of rapid focal activation in the maintenance of atrial fibrillation originating from the pulmonary veins. Pacing Clin Electrophysiol. 2000;23(11 Pt 2):1823-1827.
doi pubmed - Haissaguerre M, Jais P, Shah DC, Takahashi A, Hocini M, Quiniou G, Garrigue S, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998;339(10):659-666.
doi pubmed - Mahida S, Sacher F, Derval N, Berte B, Yamashita S, Hooks D, Denis A, et al. Science linking pulmonary veins and atrial fibrillation. Arrhythm Electrophysiol Rev. 2015;4(1):40-43.
doi pubmed - Mansour M, Ruskin J, Keane D. Initiation of atrial fibrillation by ectopic beats originating from the ostium of the inferior vena cava. J Cardiovasc Electrophysiol. 2002;13(12):1292-1295.
doi pubmed - Lin WS, Tai CT, Hsieh MH, Tsai CF, Lin YK, Tsao HM, Huang JL, et al. Catheter ablation of paroxysmal atrial fibrillation initiated by non-pulmonary vein ectopy. Circulation. 2003;107(25):3176-3183.
doi pubmed - Brunelli M, Raffa S, Grosse A, Hanazawa K, Sammut M, Roos M, Frommhold M, et al. Residual conduction after pulmonary vein isolation with a circular multielectrode radiofrequency ablation catheter: the role of adenosine and orciprenalin during a prolonged observation time. Int J Cardiol. 2013;168(4):4122-4131.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


