| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 12, Number 5, October 2021, pages 302-308
Change in Invasively Measured Mean Pulmonary Artery Pressure After Transcatheter Mitral Valve Repair Is Associated With Heart Failure Readmission
David M. Tehrania, Jiexi Wanga, Parntip Laia, Pooja S. Desaia, Heajung L. Nguyena, Lisa Banga, Eric H. Yanga, Gabriel Vorobiofa, Ali Nsaira, Olcay Aksoya, Marcella Calfon Pressa, b, Rushi V. Parikha, b, c
aDivision of Cardiology, University of California, Los Angeles, David Geffen School of Medicine, Los Angeles, CA, USA
bThese authors contributed equally to this article.
cCorresponding Author: Rushi V. Parikh, Division of Cardiology, University of California, 100 Medical Plaza, Suite 630 West, Los Angeles, CA 90095, USA
Manuscript submitted May 27, 2021, accepted July 2, 2021, published online July 28, 2021
Short title: ΔmPAP Is Associated With HF Readmission
doi: https://doi.org/10.14740/cr1284
| Abstract | ▴Top |
Background: Pre-existing pulmonary hypertension is associated with poor outcomes after transcatheter mitral valve repair (TMVr) for mitral regurgitation (MR). However, the impact of an immediate change in mean pulmonary artery pressure (ΔmPAP) following TMVr on outcomes is unknown.
Methods: Patients who underwent TMVr from December 2015 to February 18, 2020 at our institution for symptomatic 3-4+ MR and who had invasive hemodynamics measured immediately pre- and post-TMVR were included. Multivariate Cox regression analysis was performed to examine the association of ΔmPAP (post-TMVr - pre-TMVr mPAP) with the primary endpoint of heart failure (HF) readmission at 1 year. Secondary endpoints included all-cause mortality and the composite endpoint of HF readmission or all-cause mortality at 1 year.
Results: Among 55 patients, 55% were men, mean age was 72 ± 14.2 years, and mean ΔmPAP was -1.4 ± 8.2 mm Hg. Overall, HF readmission occurred in 14 (25%), death in 10 (18%), and the composite endpoint in 20 (36%) patients. In multivariable analyses, higher ΔmPAP was significantly associated with HF readmission (hazard ratio (HR) = 1.10, 95% confidence interval (CI): 1.00 - 1.21; P = 0.04). ΔmPAP was not associated with death (HR = 1.04, 95% CI: 0.96 - 1.14; P = 0.33), though there was a numerical but statistically non-significant trend towards the composite endpoint (HR = 1.06, 95% CI: 1.00 - 1.13; P = 0.06) driven by HF readmission.
Conclusion: Higher ΔmPAP immediately following TMVr was associated with increased HF readmission at 1 year. Larger prospective studies are needed to validate these data and further explore the utility of ΔmPAP as a novel hemodynamic parameter to predict post-TMVR outcomes.
Keywords: Mitral regurgitation; MitraClip; Heart failure readmission; Mortality
| Introduction | ▴Top |
Pulmonary hypertension (pHTN) due to chronic mitral regurgitation (MR) is observed in nearly one-third of patients undergoing mitral valve surgery for MR [1, 2]. Notably, pre-existing pHTN has been reported as a major risk factor for both short- and long-term all-cause mortality following mitral valve surgery [3, 4]. Over the past decade, transcatheter mitral valve repair (TMVr) using the commercially available MitraClip system (Abbot Vascular, Inc.) has been shown to be an effective intervention for patients with degenerative MR and select patients with functional MR who are deemed to be at prohibitive surgical risk [5, 6]. Similar to the surgical literature, pre-existing pHTN estimated non-invasively by transthoracic echocardiography has been associated with higher all-cause mortality among patients undergoing TMVr [7]. More recently, data from the National Cardiovascular Data Registry-based STS/ACC TVT Registry demonstrated that pHTN defined invasively prior to TMVr was associated with increased all-cause mortality and heart failure (HF) readmission at 1 year [8]. However, to the best of our knowledge, the relationship between the acute change in invasively measured following TMVr (i.e., ΔmPAP), which reflects right ventricular reserve and performance, and clinical outcomes has not been described. Therefore, in the present study, we sought to address this evidence gap and assess the association of ΔmPAP with 1-year outcomes among patients undergoing TMVr.
| Materials and Methods | ▴Top |
This retrospective cohort study included all patients from December 1, 2015 to February 18, 2020 at the University of California, Los Angeles (UCLA) who underwent TMVr using the MitraClip system for symptomatic moderate-severe (3+) or severe (4+) MR and who had invasive hemodynamics (i.e., right heart catheterization (RHC)) measured immediately before and after TMVr during the index procedure. Patients that underwent TMVr in conjunction with other procedures (e.g., transcatheter aortic valve placement or percutaneous tricuspid valve repair) were excluded. This study was approved by the UCLA institutional review board. This study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
Procedural success was defined as improvement of ≥ 2+ MR after TMVr based on intraprocedural transesophageal echocardiogram. Post-mPAP pressures were obtained after TMVr was complete and the patient had stable pulmonary pressures for at least 5 min. We defined ΔmPAP as post-TMVr mPAP - pre-TMVr mPAP. The primary endpoint was HF readmission at 1 year. Secondary endpoints included all-cause mortality at 1 year and the composite of HF readmission or all-cause mortality at 1 year. All HF readmissions were reviewed and adjudicated by two authors (David M. Tehrani and Rushi V. Parikh).
Data are expressed as frequency (percentage) or mean ± standard deviation. Non-parametric Wilcoxon rank-sum or rank-sign tests were used to evaluate differences between groups of variables, as appropriate. Baseline demographic, cardiovascular, and procedural characteristics were tested for their ability to predict the primary and secondary endpoints using univariable Cox proportional hazards regression models. Covariates with a univariate P-value < 0.10 were included in multivariable models to assess for independent predictors. Cox regression data are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). Statistical analyses were performed using the SPSS system, version 26 (SPSS Inc., Chicago, IL). A P-value < 0.05 was considered statistically significant.
| Results | ▴Top |
A total of 116 patients received TMVr at UCLA from December 1, 2015 to February 18, 2020, 55 of whom met the outlined inclusion criteria and were included in the present study. Sixty-one patients were excluded for the following reasons: 49 did not undergo both pre- and post-TMVr RHC and 12 underwent concomitant procedures at the time of TMVr.
Among the 55 patients comprising the study cohort, the mean age was 72 ± 14 years, 55% were men, 64% had degenerative MR, and 84% had 4+ MR. Procedural characteristics included 91% with successful TMVr, a mean post-TMVr mitral gradient of 3.9 ± 1.4 mm Hg, and 1.7 ± 0.76 MitraClips used. Overall, the mean pre-mPAP was 31.5 ± 10.1 mm Hg, post-mPAP was 30.1 ± 7.4 mm Hg, and ΔmPAP was -1.4 ± 8.2 mm Hg (Table 1). The mean ΔmPAP in those free of HF readmission at 1 year was -3.0 ± 8.5 mm Hg and in those with a HF readmission at 1 year was 3.1 ± 5.2 mm Hg (Fig. 1). Notably, there was no significant difference in the use of guideline-directed medical therapy (e.g., beta-blockers, angiotensin converting enzyme inhibitors or angiotensin receptor blockers, aldosterone antagonists, and loop diuretics) between the HF readmission cohort and those without HF readmission.
 Click to view | Table 1. Baseline Clinical, Echocardiographic, and Procedural Characteristics |
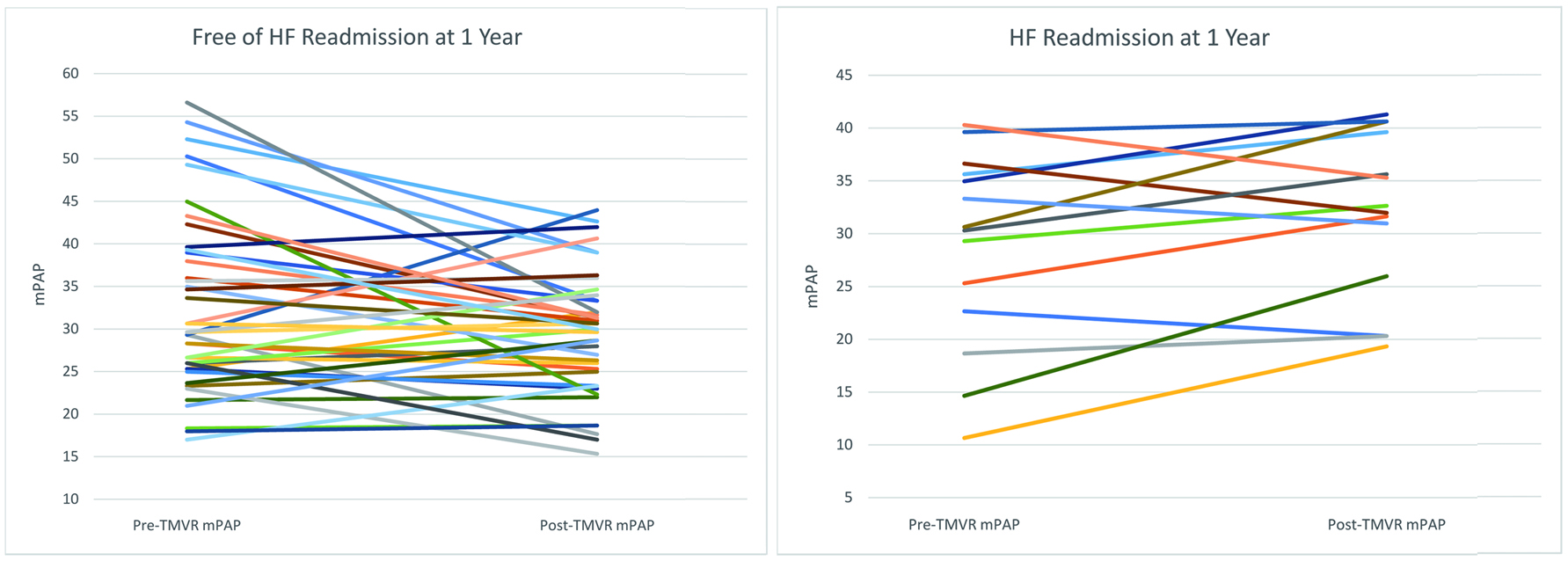 Click for large image | Figure 1. Immediate change in invasively measured mPAP after TMVr. Patients free from HF readmission (a) and those with a HF readmission (b) at 1 year are depicted with their immediate pre- and post-TMVr mPAP after undergoing TMVr. HF: heart failure; mPAP: mean pulmonary artery pressure; TMVr: transcatheter mitral valve repair. |
In univariable Cox regression analyses, elevated ΔmPAP (HR = 1.10, 95% CI: 1.01 - 1.19; P = 0.03) and atrial fibrillation (HR = 0.09, 95% CI: 0.11 - 1.16; P = 0.09) were associated with HF readmission (P < 0.10) and included in the multivariable model (Table 2). After adjustment, elevated ΔmPAP remained as the only independent predictor of HF readmission (HR = 1.10, 95% CI: 1.00 - 1.21; P = 0.04) (Table 2). Higher ΔmPAP was not significantly associated with all-cause mortality at 1 year in univariable analysis (HR = 1.04, 95% CI: 0.96 - 1.14; P = 0.33), while procedural success was associated with freedom from death at 1 year (HR = 0.11, 95% CI: 0.03 - 0.41; P < 0.01 (Table 3). However, driven by HF readmission, elevated ΔmPAP demonstrated a numerical but statistically non-significant association with the composite endpoint of HF readmission or all-cause mortality at 1 year in multivariable analyses (HR = 1.06, 95% CI: 1.00 - 1.13; P = 0.06). Similarly, in multivariable analysis, procedural success had a modest trend towards statistical significance for reduction in the composite endpoint (HR = 0.37, 95% CI: 0.12 - 1.11; P = 0.08) (Table 4).
 Click to view | Table 2. Association of Clinical and Procedural Characteristics With Heart Failure Readmission at 1 Year |
 Click to view | Table 3. Association of Clinical and Procedural Characteristics With All-Cause Mortality at 1 Year |
 Click to view | Table 4. Association of Clinical and Procedural Characteristics With Heart Failure Readmission or All-Cause Mortality at 1 Year |
| Discussion | ▴Top |
The principal finding of this retrospective cohort study of patients undergoing TMVr using the MitraClip system for symptomatic 3-4+ MR was that elevated ΔmPAP (i.e., immediate increase in mPAP following TMVr) was significantly associated with HF readmission at 1 year. Our study builds upon previously reported peri-TMVr invasive hemodynamic data and highlights the impact of pulmonary pressures and real-time hemodynamic changes on outcomes in patients undergoing TMVr.
Recent data from the STS/ACC TVT Registry among patients undergoing TMVr demonstrated a significant relationship between escalating degrees of invasively defined pHTN prior to TMVr with increased HF readmission and all-cause mortality at 1 year regardless of MR etiology [8]. However, important limitations of these registry-based data are: 1) the pre-TMVr invasive hemodynamic assessment via RHC was not uniformly done during the index procedure, and 2) post-TMVr hemodynamic data were not captured. In the present study, we addressed these gaps and found that higher ΔmPAP immediately after TMVr was significantly associated with increased HF readmission at 1 year. To the best of our knowledge, the impact of immediate changes in pulmonary arterial hemodynamics after TMVr on outcomes has not been previously described. These novel hypothesis-generating data suggest that ΔmPAP may provide real-time prognostic value and add to the growing evidence that pulmonary arterial hemodynamics appear to significantly contribute to TMVr outcomes.
The immediate improvement/reversibility in pulmonary pressures following TMVr (i.e., low ΔmPAP) may indicate greater right ventricle (RV) functional reserve and/or performance. Whether this hemodynamic malleability reflects a lesser extent and/or chronicity of pulmonary vascular remodeling requires further investigation. Pulmonary vascular pathophysiology in the setting of chronic MR is complex, characterized by early pulmonary venous hypertension followed by a later precapillary component driven by structural/functional changes in the pulmonary arterial vasculature, ultimately leading to elevated pulmonary vascular resistance [9]. Taken together, it is conceivable that patients with chronic MR and irreversible pHTN may not benefit as greatly from TMVr compared with their counterparts who are able to achieve meaningful reductions in pulmonary pressures. Indeed, procedural success did not necessarily result in an immediate drop in pulmonary pressures in our study, suggesting that chronic pulmonary vascular remodeling may contribute to adverse outcomes despite procedural success.
Obtaining post-TMVr invasive hemodynamics is not standard practice, and as such is not captured by the STS/ACC TVT registry. Nonetheless, there are emerging data suggesting that continuous hemodynamic monitoring during TMVr may predict improvement in clinical outcomes, such as post-TMVr improvement in left atrial pressure predicting improvement in the 6-min walk test [10]. In contrast, post-TMVr echocardiographic parameters such as procedural success are required data elements of the registry. Unsurprisingly, procedural success has been associated with improved outcomes, and we similarly observed that procedural success was associated with decreased all-cause mortality [11]. However, given that unsuccessful TMVr procedures are uncommon in the contemporary era given improved operator experience and device technology, additional procedural markers of outcomes are needed in patients with successful TMVr. Our data suggest that immediate improvement in pulmonary pressures after TMVr (i.e., ΔmPAP) may be a potential candidate hemodynamic parameter worthy of further investigation.
This study has multiple limitations worth considering. First, this is a single center retrospective study with a modest TMVr sample size, and thus the data are hypothesis-generating and require prospective validation. Second, the non-consecutive nature of our cohort may have introduced selection bias affecting the reported observations, though differences between demographics and procedural characteristics in the study and excluded cohorts were limited. Third, despite adjusting for known patient and procedural confounders, we cannot rule out residual confounding (e.g., hemodynamic loading conditions, respiratory variation, etc.) prior to the TMVr. Fourth, given the retrospective nature of our study, data for other key hemodynamic predictors such as change in left atrial pressure were incomplete/limited. Finally, although outcomes data were adjudicated based on extensive chart review rather than administrative claims data, which provided incremental rigor, HF readmissions at institutions outside of UCLA were not captured.
Conclusion
In summary, an immediate rise in invasively measured mPAP following TMVr (i.e., high ΔmPAP) was significantly associated with increased HF readmission at 1 year. This hemodynamic parameter may help risk stratify patients early after TMVr irrespective of procedural success and guide personalized follow-up care. Larger, prospective, multicenter studies are needed to confirm these hypothesis-generating data and determine the utility of ΔmPAP as a predictor of outcomes post-TMVr.
Acknowledgments
None to declare.
Financial Disclosure
This research received no grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest
MCP is a proctor for the Abbott MitraClip Procedure. No other authors have relevant disclosures. RVP reports unrelated activities including research support from the American Heart Association, consulting fees from Abbott, and serving on the scientific advisory board (minor equity interest) of Stallion Cardio, DocVocate, and HeartCloud.
Informed Consent
Informed consent was waived for this retrospective cohort study.
Author Contributions
DMT contributed to study design, engaged in data collection, analyzed and interpreted the data, and drafted the manuscript. JW engaged in data collection, analyzed and interpreted the data, and drafted the manuscript. PL, HLN, LB, and PSD engaged in data collection and contributed to drafts of the manuscript. EHY, GV, AN, and OA interpreted the data and provided critical reviews of the manuscript. MCP and RVP designed the study, analyzed and interpreted data, and provided critical review of the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
CI: confidence interval; HF: heart failure; HR: hazard ratio; MR: mitral regurgitation; pHTN: pulmonary hypertension; RHC: right heart catheterization; RV: right ventricle; SPSS: Statistical Package for the Social Sciences; TMVr: transcatheter mitral valve repair; UCLA: University of California, Los Angeles; ΔmPAP: mean pulmonary artery pressure
| References | ▴Top |
- Vachiery JL, Adir Y, Barbera JA, et al. Pulmonary hypertension due to left heart diseases. Journal of the American College of Cardiology. 2013;62:25.
doi pubmed - Maeder MT, Weber L, Buser M, Gerhard M, Haager PK, Maisano F, Rickli H. Pulmonary hypertension in aortic and mitral valve disease. Front Cardiovasc Med. 2018;5:40.
doi pubmed - Mentias A, Patel K, Patel H, Gillinov AM, Sabik JF, Mihaljevic T, Suri RM, et al. Effect of pulmonary vascular pressures on long-term outcome in patients with primary mitral regurgitation. J Am Coll Cardiol. 2016;67(25):2952-2961.
doi pubmed - Ghoreishi M, Evans CF, DeFilippi CR, Hobbs G, Young CA, Griffith BP, Gammie JS. Pulmonary hypertension adversely affects short- and long-term survival after mitral valve operation for mitral regurgitation: implications for timing of surgery. J Thorac Cardiovasc Surg. 2011;142(6):1439-1452.
doi pubmed - Kar S, Feldman T, Qasim A, Trento A, Kapadia S, Pedersen W, Lim DS, et al. Five-year outcomes of transcatheter reduction of significant mitral regurgitation in high-surgical-risk patients. Heart. 2019;105(21):1622-1628.
doi pubmed - Stone GW, Lindenfeld J, Abraham WT, Kar S, Lim DS, Mishell JM, Whisenant B, et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018;379(24):2307-2318.
doi pubmed - Matsumoto T, Nakamura M, Yeow WL, Hussaini A, Ram V, Makar M, Gurudevan SV, et al. Impact of pulmonary hypertension on outcomes in patients with functional mitral regurgitation undergoing percutaneous edge-to-edge repair. Am J Cardiol. 2014;114(11):1735-1739.
doi pubmed - Al-Bawardy R, Vemulapalli S, Thourani VH, Mack M, Dai D, Stebbins A, Palacios I, et al. Association of pulmonary hypertension with clinical outcomes of transcatheter mitral valve repair. JAMA Cardiol. 2020;5(1):47-56.
doi pubmed - Patel H, Desai M, Tuzcu EM, Griffin B, Kapadia S. Pulmonary hypertension in mitral regurgitation. J Am Heart Assoc. 2014;3(4):e000748.
doi - Maor E, Raphael CE, Panaich SS, Reeder GS, Nishimura RA, Nkomo VT, Rihal CS, et al. Acute changes in left atrial pressure after mitraclip are associated with improvement in 6-minute walk distance. Circ Cardiovasc Interv. 2017;10(4):e004856.
doi - Feldman T, Foster E, Glower DD, Kar S, Rinaldi MJ, Fail PS, Smalling RW, et al. Percutaneous repair or surgery for mitral regurgitation. N Engl J Med. 2011;364(15):1395-1406.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


