| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Short Communication
Volume 12, Number 6, December 2021, pages 358-362
A Significant Increase of Estimated Glomerular Filtration Rate After Switching From Fenofibrate to Pemafibrate in Type 2 Diabetic Patients
Hidekatsu Yanaia, b, Hisayuki Katsuyamaa, Mariko Hakoshimaa
aDepartment of Diabetes, Endocrinology and Metabolism, National Center for Global Health and Medicine Kohnodai Hospital, Chiba, Japan
bCorresponding Author: Hidekatsu Yanai, Department of Diabetes, Endocrinology and Metabolism, National Center for Global Health and Medicine Kohnodai Hospital, 1-7-1 Kohnodai, Ichikawa, Chiba 272-8516, Japan
Manuscript submitted November 2, 2021, accepted November 16, 2021, published online November 29, 2021
Short title: eGFR in Type 2 Diabetic Patients
doi: https://doi.org/10.14740/cr1333
| Abstract | ▴Top |
Background: Dyslipidemia is one of the major risk factors for cardiovascular disease (CVD), along with hypertension, diabetes, smoking and obesity. Approximately 70% of CVD risk remains even after treatment of elevated low-density lipoprotein-cholesterol (LDL-C) by statins. High triglyceride (TG) and low high-density lipoprotein-cholesterol (HDL-C) level are potential therapeutic targets to prevent CVD. Fibrates were associated with a greater reduction in TG, and a greater increase in HDL-C. Fibrates activate specific transcription factors belonging to the nuclear hormone receptor superfamily, termed peroxisome proliferator-activated receptors (PPARs). Fibrates improve atherogenic dyslipidemia by mediating PPARα. Pemafibrate is a novel member of the selective PPARα modulator (SPPARMα) family that was designed to have a higher PPARα agonistic activity and selectivity than previous fibrates. Here, we aimed to study the influences of the switching from fenofibrate to pemafibrate on metabolic parameters in type 2 diabetic patients.
Methods: We retrospectively picked up type 2 diabetic patients who had undergone the switching from fenofibrate to pemafibrate, and compared metabolic parameters before the switching with the data at 3, 6 and 12 months after the switching.
Results: We found 15 patients with type 2 diabetes. Serum alanine aminotransferase significantly decreased at 6 months after the switching as compared with baseline. The estimated glomerular filtration rate (eGFR) significantly increased at 3, 6 and 12 months after the switching from fenofibrate to pemafibrate as compared with baseline. Serum uric acid (UA) levels significantly increased at 3 and 6 months after the switching as compared with baseline. We did not observe changes in other metabolic parameters after the switching.
Conclusion: We observed a significant increase of eGFR and serum UA after the switching from fenofibrate to pemafibrate in type 2 diabetic patients. Recent evidences suggest that the improvement of eGFR is beneficially associated with the development of CVD in type 2 diabetic patients. Considering the impact on eGFR, pemafibrate may effectively reduce CVD as compared with fenofibrate.
Keywords: Cardiovascular disease; Estimated glomerular filtration rate; Fenofibrate; Pemafibrate; Uric acid
| Introduction | ▴Top |
Dyslipidemia is one of the major risk factors for atherosclerotic cardiovascular disease (ASCVD), along with hypertension, diabetes, smoking and obesity. The meta-analysis showed that statins reduce the risk of ASCVD by approximately 20-30%, suggesting that approximately 70% of ASCVD risk remains even after treatment of high low-density lipoprotein-cholesterol (LDL-C) by statins [1, 2]. High levels of triglyceride (TG)-rich lipoproteins such as intermediate-density lipoprotein (IDL) and very-low-density lipoprotein (VLDL) and low high-density lipoprotein cholesterol (HDL-C) level are commonly observed in patients with type 2 diabetes and obesity [3, 4]. Such atherogenic dyslipidemias are potential therapeutic targets to prevent ASCVD.
An optimal reduction of cardiovascular risk through the management of atherogenic dyslipidemias depends on efficacy of lipid-modulating agents beyond statin-based reduction of LDL-C. The most important class of medications to manage atherogenic dyslipidemias can be fibrates, because fibrates were associated with a greater reduction in TG, and a greater increase in HDL-C [5]. Fibrates activate specific transcription factors belonging to the nuclear hormone receptor superfamily, termed peroxisome proliferator-activated receptors (PPARs) [6]. Three isoforms are identified: PPARα, PPARγ and PPARβ/δ [7]. PPARα is abundant in energy-demanding tissues, such as the liver, kidney, heart and skeletal muscle; PPARγ is predominantly found in adipose tissue, macrophages and the large intestine, whereas PPARβ/δ is more ubiquitous in distribution [8, 9]. These PPARs are controlled through their interaction with fatty acids (FAs) and their derivatives and are the pharmacological targets for fibrates (PPARα) or the insulin sensitizer thiazolidinediones (PPARγ). Fibrates elevate HDL-C levels via transcriptional induction of synthesis of HDL apolipoproteins by mediating PPARα. By mediating PPARα, fibrates also reduce serum TG by lowering hepatic apo C-III production and by increasing lipoprotein lipase-mediated lipolysis and FA oxidation.
Pemafibrate is a novel member of the selective PPARα modulator (SPPARMα) family that was designed to have a higher PPARα agonistic activity and selectivity than existing PPARα agonists (such as fibrates) [8, 10]. Here, we aimed to study the influences of the switching from fenofibrate to pemafibrate on metabolic parameters in type 2 diabetic patients.
| Materials and Methods | ▴Top |
We retrospectively picked up type 2 diabetic patients who had undergone the switching from fenofibrate to pemafibrate, and compared metabolic parameters before the switching with the data at 3, 6 and 12 months after the switching. The study protocol was approved by the Ethics Committee of the National Center for Global Health and Medicine, and the study was performed in accordance with the Declaration of Helsinki. The paired t-test was used to statistically analyze comparison in metabolic parameters between before and after the switching.
| Results | ▴Top |
We found 15 patients with type 2 diabetes. Baseline characteristics for type 2 diabetic patients who had undergone the switching from fenofibrate to pemafibrate are shown in Table 1. Ten patients had hypertension, nine patients were overweight, and eight patients had received non-fibrate lipid-lowering drugs.
 Click to view | Table 1. Characteristics for Type 2 Diabetic Patients Who Had Undergone the Switching From Fenofibrate to Pemafibrate (n = 15 |
Clinical and biochemical parameters except for estimated glomerular filtration rate (eGFR) and serum uric acid (UA) of patients before and after the switching are shown in Table 2. Only serum alanine aminotransferase significantly decreased at 6 months after the switching as compared with baseline. Other parameters did not show a significant change after the switching. Changes in eGFR and serum UA are shown in Figure 1. The eGFR significantly increased at 3, 6 and 12 months after the switching from fenofibrate to pemafibrate as compared with baseline. Serum UA levels significantly increased at 3 and 6 months after the switching as compared with baseline.
 Click to view | Table 2. Changes in Metabolic Parameters After Switching From Fenofibrate to Pemabibrate |
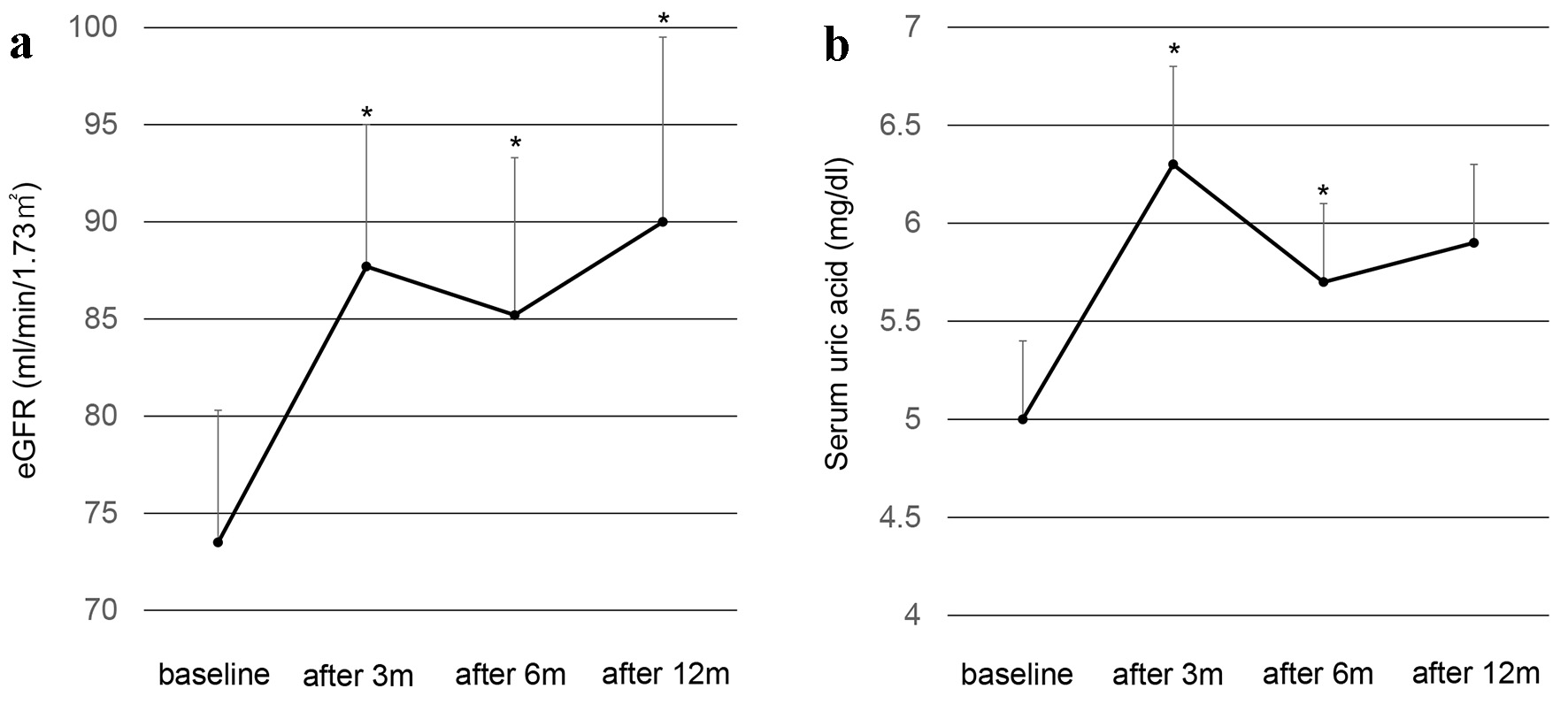 Click for large image | Figure 1. Changes in eGFR (a) and serum uric acid (b) after the switching from fenofibrate to pemafibrate in type 2 diabetic patients. Black circles and error bars indicate mean and SD, respectively. *P < 0.05 vs. values at baseline. eGFR: estimated glomerular filtration rate; SD: standard deviation. |
| Discussion | ▴Top |
The lipid profile of patients with type 2 diabetes, obesity and metabolic syndrome is characterized by low HDL-C levels and increased TG [3, 4], which has been defined as atherogenic dyslipidemia. Fibrates can be effectively used to manage such atherogenic dyslipidemias. Randomized control trials showed that gemfibrozil, fenofibrate and bezafibrate were significantly effective to prevent CVD events in patients with atherogenic dyslipidemia [11]. In the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study, fenofibrate significantly reduced CVD events in those with low HDL-C or hypertension [12]. The largest effect of fenofibrate to reduce CVD risk was observed in subjects with marked dyslipidemia (TG ≥ 2.3 mmol/L and low HDL-C) in whom a 27% relative risk reduction (95% confidence interval: 9 - 42; P = 0.005) was observed [12]. Different fibrates may have different spectrum of beneficial effects and adverse effects.
In our study, serum UA significantly increased after the switching from fenofibrate to pemafibrate. Fenofibrate influences on serum UA metabolism [13]. Fenofibrate was reported to decrease serum UA levels by increasing its urinary excretion, most likely through the inhibition of urate transporter 1 (URAT1) by fenofibric acid, its major metabolite [14]. The meta-analyses also demonstrated that fenofibrate significantly reduced serum UA levels [15, 16]. However, reduction of serum UA by pemafibrate was not reported, suggesting that fenofibrate has URAT1 inhibitory effect; however, pemafibrate may not have such effect. The URAT1 inhibitory effect by pemafibrate should be studied in the future.
In our study, eGFR significantly increased after the switching from fenofibrate to pemafibrate. Fenofibrate was reported to increase serum creatinine levels and decrease the eGFR [17]. In the FIELD Helsinki sub-study, a significant decrease in eGFR was observed in the fenofibrate group [18]. The mechanism of fibrate-induced renal function impairment is unclear. Potential mechanisms include increased muscular production of creatinine, decreased secretion from renal tubules, and a change in the glomerular filtration through altered hemodynamics. Hottelart et al postulated that increased serum creatinine levels result from increased creatinine production [19]. Fibrates reduce the production of vasodilatory prostaglandins, leading to a change in the renal function in patients who experience a rise in the serum creatinine levels [20], which is another possible explanation.
In the multicenter, single-arm, open-label, phase III trial, 0.2 - 0.4 mg/day pemafibrate was administered for 52 weeks to 189 patients with hypertriglyceridemia and an eGFR ≥ 45 mL/min/1.73 m2 on statin or regardless of eGFR when statin was not administered [21]. There were no significant changes in eGFR over time in any stages of chronic kidney disease (CKD) [21]. Further, pemafibrate showed a good safety profile and efficacy in correcting lipid abnormalities in a broad range of patients, including those with CKD. Fenofibrate is metabolized mainly by the kidneys, whereas pemafibrate is metabolized mainly by the liver [22], which can partially explain a significant increase of eGFR after the switching from fenofibrate to pemafibrate. In an on-drug/off-drug ancillary study to the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Lipid Trial to investigate posttrial changes in serum creatinine, participants with significant initial on-trial increases in serum creatinine (≥ 20%) returned to the same level of renal function as participants receiving placebo, suggesting that the fenofibrate-associated on-trial increases in serum creatinine were reversible [23]. A significant increase of eGFR after the switching from fenofibrate to pemafibrate in our study may reflect the effect of discontinuation of fenofibrate. It remains unknown whether pemafibrate improves eGFR or not in type 2 diabetic patients, which should be studies in the future.
Recent studies using sodium glucose cotransporter 2 inhibitors (SGLT2is) demonstrates that the improvement of eGFR is beneficially associated with the development of CVD in type 2 diabetic patients [24, 25], suggesting a significance of effects of drugs on eGFR when managing cardiovascular risks in patients with type 2 diabetes. Considering the impact on eGFR, pemafibrate may be more beneficially associated with the development of CVD as compared with fenofibrate.
Limitations of the study need to be addressed. This is a cross-sectional study, limiting inferences of causality and its direction. Although treatments for diabetes and hypertension and other lipid-lowering drugs did not change during the study period, we cannot deny that such treatment might influence on the change of eGFR. We should mention further limitations on the small sample size. Further studies, preferably with larger numbers of subjects, will be needed in the future.
In conclusion, we observed a significant increase of eGFR and serum UA after the switching from fenofibrate to pemafibrate in type 2 diabetic patients.
Acknowledgments
We thank the staffs of the Division of Research Support, National Center for Global Health and Medicine Kohnodai Hospital.
Financial Disclosure
Authors have no financial disclosures to report.
Conflict of Interest
The authors declare that they have no conflict of interest concerning this article.
Informed Consent
Not applicable.
Author Contributions
HY designed the research, and MH and HK collected and analyzed data. HY wrote and approved the final paper.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Cholesterol Treatment Trialists' (CTT) Collaboration, Fulcher J, O'Connell R, Voysey M, Emberson J, Blackwell L, Mihaylova B, et al. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet. 2015;385(9976):1397-1405.
doi - Chapman MJ, Redfern JS, McGovern ME, Giral P. Niacin and fibrates in atherogenic dyslipidemia: pharmacotherapy to reduce cardiovascular risk. Pharmacol Ther. 2010;126(3):314-345.
doi pubmed - Yanai H, Hirowatari Y, Ito K, Kurosawa H, Tada N, Yoshida H. Understanding of diabetic dyslipidemia by using the anion-exchange high performance liquid chromatography data. J Clin Med Res. 2016;8(5):424-426.
doi pubmed - Yanai H, Hirowatari Y, Yoshida H. Diabetic dyslipidemia: evaluation and mechanism. Glob Health Med. 2019;1(1):30-35.
doi pubmed - Abourbih S, Filion KB, Joseph L, Schiffrin EL, Rinfret S, Poirier P, Pilote L, et al. Effect of fibrates on lipid profiles and cardiovascular outcomes: a systematic review. Am J Med. 2009;122(10):962 e961-968.
doi pubmed - Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation. 1998;98(19):2088-2093.
doi pubmed - Issemann I, Green S. Activation of a member of the steroid hormone receptor superfamily by peroxisome proliferators. Nature. 1990;347(6294):645-650.
doi pubmed - Fruchart JC. Selective peroxisome proliferator-activated receptor alpha modulators (SPPARMalpha): the next generation of peroxisome proliferator-activated receptor alpha-agonists. Cardiovasc Diabetol. 2013;12:82.
doi pubmed - Braissant O, Foufelle F, Scotto C, Dauca M, Wahli W. Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology. 1996;137(1):354-366.
doi pubmed - Yamazaki Y, Abe K, Toma T, Nishikawa M, Ozawa H, Okuda A, Araki T, et al. Design and synthesis of highly potent and selective human peroxisome proliferator-activated receptor alpha agonists. Bioorg Med Chem Lett. 2007;17(16):4689-4693.
doi pubmed - Tenenbaum A, Fisman EZ. Fibrates are an essential part of modern anti-dyslipidemic arsenal: spotlight on atherogenic dyslipidemia and residual risk reduction. Cardiovasc Diabetol. 2012;11:125.
doi pubmed - Scott R, O'Brien R, Fulcher G, Pardy C, D'Emden M, Tse D, Taskinen MR, et al. Effects of fenofibrate treatment on cardiovascular disease risk in 9,795 individuals with type 2 diabetes and various components of the metabolic syndrome: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetes Care. 2009;32(3):493-498.
doi pubmed - Yanai H, Adachi H, Hakoshima M, Katsuyama H. Molecular biological and clinical understanding of the pathophysiology and treatments of hyperuricemia and its association with metabolic syndrome, cardiovascular diseases and chronic kidney disease. Int J Mol Sci. 2021;22(17):9221.
doi pubmed - Uetake D, Ohno I, Ichida K, Yamaguchi Y, Saikawa H, Endou H, Hosoya T. Effect of fenofibrate on uric acid metabolism and urate transporter 1. Intern Med. 2010;49(2):89-94.
doi pubmed - Zhang J, Ji X, Dong Z, Lu J, Zhao Y, Li R, Li C, et al. Impact of fenofibrate therapy on serum uric acid concentrations: a review and meta-analysis. Endocr J. 2021;68(7):829-837.
doi pubmed - Derosa G, Maffioli P, Sahebkar A. Plasma uric acid concentrations are reduced by fenofibrate: A systematic review and meta-analysis of randomized placebo-controlled trials. Pharmacol Res. 2015;102:63-70.
doi pubmed - Kim S, Ko K, Park S, Lee DR, Lee J. Effect of Fenofibrate Medication on Renal Function. Korean J Fam Med. 2017;38(4):192-198.
doi pubmed - Forsblom C, Hiukka A, Leinonen ES, Sundvall J, Groop PH, Taskinen MR. Effects of long-term fenofibrate treatment on markers of renal function in type 2 diabetes: the FIELD Helsinki substudy. Diabetes Care. 2010;33(2):215-220.
doi pubmed - Hottelart C, El Esper N, Rose F, Achard JM, Fournier A. Fenofibrate increases creatininemia by increasing metabolic production of creatinine. Nephron. 2002;92(3):536-541.
doi pubmed - Sica DA. Fibrate therapy and renal function. Curr Atheroscler Rep. 2009;11(5):338-342.
doi pubmed - Yokote K, Yamashita S, Arai H, Araki E, Suganami H, Ishibashi S, Of The KSGOB. Long-term efficacy and safety of pemafibrate, a novel selective peroxisome proliferator-activated receptor-alpha modulator (SPPARMalpha), in dyslipidemic patients with renal impairment. Int J Mol Sci. 2019;20(3):706.
doi pubmed - Yamashita S, Masuda D, Matsuzawa Y. Clinical applications of a novel selective PPARalpha modulator, pemafibrate, in dyslipidemia and metabolic diseases. J Atheroscler Thromb. 2019;26(5):389-402.
doi pubmed - Mychaleckyj JC, Craven T, Nayak U, Buse J, Crouse JR, Elam M, Kirchner K, et al. Reversibility of fenofibrate therapy-induced renal function impairment in ACCORD type 2 diabetic participants. Diabetes Care. 2012;35(5):1008-1014.
doi pubmed - Yanai H, Hakoshima M, Adachi H, Katsuyama H. Multi-organ protective effects of sodium glucose cotransporter 2 inhibitors. Int J Mol Sci. 2021;22(9):4416.
doi pubmed - Yanai H. Acute effects of preventing heart failure by sodium-glucose cotransporter 2 inhibitors. Cardiol Res. 2021;12(5):324-326.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


