| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 13, Number 3, June 2022, pages 135-143
Bradyarrhythmia After Remdesivir Administration in SARS-CoV-2: A Review of Literature and Meta-Analysis of Observational Studies in Epidemiology
Safa Al-Jammalia, d, Rana Al-Zakharia, Nicholas Sheetsb, d, Arun Mahtania, Veronika Stefanishinab, Nidal Isberc
aDepartment of Medicine, Richmond University Medical Center, Staten Island, NY, USA
bSt. George’s University School of Medicine, Grenada, West Indies
cDepartment of Electrophysiology, Richmond University Medical Center, Staten Island, NY, USA
dCorresponding Author: Safa Al-Jammali, Department of Medicine, Richmond University Medical Center, Staten Island, NY, USA; Nicholas Sheets, St. George’s University School of Medicine, Grenada, West Indies
Manuscript submitted March 29, 2022, accepted April 13, 2022, published online May 10, 2022
Short title: Bradyarrhythmia After Remdesivir in COVID-19
doi: https://doi.org/10.14740/cr1377
| Abstract | ▴Top |
Background: The coronavirus disease 2019 (COVID-19) pandemic has required timely and informed decisions about treatment recommendations for clinical practice. One such drug used for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is remdesivir (RDV), and several cardiac side effects have been reported including bradyarrhythmia (e.g., transient sinus bradycardia, symptomatic sinus bradycardia, complete atrioventricular (AV) block). The current study aimed to explore the association between RDV treatment for SARS-CoV-2 infection and the risk of bradyarrhythmia by presenting a review and meta-analysis of available published studies.
Methods: We presented a review of published literature and meta-analysis of observational studies (MOOSE). A narrative summary of RDV and bradyarrhythmia in COVID-19 infection and pooled analysis of observational studies that meet inclusion criteria was included. Studies included were published between January 2020 and December 2021 (identified through PubMed and ScienceDirect) and examined the association between treatment with RDV in SARS-CoV-2 infection and the risk of bradyarrhythmia.
Results: Three studies (two retrospective cohort studies and one prospective cohort study) met inclusion criteria for pooled meta-analysis of bradyarrhythmia and RDV therapy in COVID-19 patients. Treatment with RDV was associated with increased risk of sinus bradycardia when compared to controls (odds ratio 3.27 (95% confidence interval 1.90 - 5.63)). In the pooled analysis, the incidence of bradycardia in those that received RDV was 34.07% vs. 18.13% among controls. Thirteen case reports, three case series, and three disproportionality analyses were identified in review of the literature.
Conclusion: Data from real-world observational studies suggest that treating COVID-19 patients with RDV may predispose the development of bradyarrhythmia. The importance of this observation is of uncertain clinical significance as some observational studies have reported more favorable outcomes among patients who experience bradycardia after RDV therapy. The current study is limited by the small number of studies that could be meaningfully pooled and more well-designed cohort studies are needed to explore this association.
Keywords: Meta-analysis; Bradyarrhythmia; Sinus bradycardia; Remdesivir: COVID-19; Observational studies
| Introduction | ▴Top |
Since December 2019, the collaborative efforts of the medical and scientific community have been determined to identify, manage, and treat coronavirus disease 2019 (COVID-19) that emerged from Wuhan City, Hubei Province, Central China, as cases of pneumonia with unknown etiology, later identified as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), have led to a global pandemic [1]. SARS-CoV-2 belongs to the family Coronaviridae and possesses a single-strand, positive-sense RNA genome [2]. Presently, the only antiviral drug approved in the United States by the Food and Drug Administration (FDA) is remdesivir (RDV), which obtained Emergency Authorization Use (EAU) on May 1, 2020 (modified on August 28, 2020) after preliminary results were released from Adaptive COVID-19 Treatment Trial (ACTT-1) [3]. ACTT-1 included a total of 1,062 patients that were randomized (541 to RDV and 521 to placebo) and demonstrated a median recovery time of 10 days (95% confidence interval (CI) 9 - 11) for the RDV group compared with 15 days (95% CI 13 - 18) in the placebo group [3]. RDV (GS-5734) inhibits viral RNA-dependent RNA polymerase, which is a structurally conserved enzyme integral in the replication of a wide range of viruses, including Coronaviridae [3, 4]. Despite promising data from the randomized controlled trial that led to the EUA, RDV has exhibited noteworthy adverse cardiovascular side effects in pharmacovigilance studies. A study using VigiBase found 302 cardiac effects from the administration of RDV among 2,603 adverse events, with 31% (N = 94) being bradycardia [5]. When compared with other potential treatments for COVID-19 (e.g., hydroxychloroquine, lopinavir/ritonavir, tocilizumab, or glucocorticoids), RDV was associated with an increased risk of reporting bradycardia (reporting odds ratio (ROR) 1.65; 95% CI 1.23 - 2.22) [5].
The rapid nature of COVID-19 pandemic has called for timely and informed decisions for clinical practice and public health efforts. The current study aimed to explore the association between RDV treatment in the setting of SARS-CoV-2 infection and the risk of bradyarrhythmia. To accomplish this, we presented a review of the literature and a meta-analysis of real-world data from observational studies. The current study used a pooled analysis of observational studies that measure the association between bradyarrhythmia and the use of RDV in COVID-19-infected individuals. Additionally, a narrative review of case reports, case series, and observational studies identified in literature of bradyarrhythmia after RDV administration was included.
| Materials and Methods | ▴Top |
Study selection
Selection of studies was performed with PubMed and ScienceDirect to search for studies that reported bradyarrhythmia in patients with COVID-19 infection taking RDV published up December 2021 with the following search terms: “bradycardia” or “bradyarrhythmia” in combination with “SARS-CoV-2” or “COVID-19” and “remdesivir” with no restrictions. References and abstracts of potential studies were reviewed. Case reports, case series, and editorials were excluded from pooled analysis, however, included in the review of literature.
Inclusion and exclusion criteria
If the study met the following conditions, it should be included: patients should meet the diagnostic criteria for bradycardia for those reporting bradycardia, subjects should be treated with RDV, a control group should be included in study design, the controls should be treated with other therapies or no other treatment, the study should be published in English, and available data should be extracted from the article or obtained by calculation.
Data collection
We conducted data collection in a standardized manner. The following was collected from the included studies: first author’s last name, publication year, study population, size of the sample, and outcome variables. Because the data included in this study were retrieved from the literature, IRB approval was not needed.
Bias and quality assessment
Studies included in the current study were observational and the quality was assessed using the Newcastle-Ottawa Quality Assessment scale for meta-analysis. All of the above procedures were performed by two independent authors and any disagreements were resolved with discussion of data quality and inclusion/exclusion criteria.
Statistical analysis
Statistical analysis was performed using R studio version 1.4.1. The meta-package was used to perform meta-analyses. Metabin function was used to calculate odds ratio (OR) of developing bradyarrhythmia in patients that received RDV and in patients that did not in three separate studies. A forest plot was created using the forest function to visualize the results. Publication bias was measured using a funnel plot and Egger test.
| Results | ▴Top |
Study selection process and study characteristics
Selection of studies was performed with PubMed and ScienceDirect databases using the keywords “bradycardia,” “bradyarrhythmia,” “COVID-19,” “SARS-CoV-2,” and “remdesivir” and Boolean string functions (e.g., AND, OR). Articles identified from initial search (PubMed, n = 28, ScienceDirect, n = 115) were evaluated via abstract and title for exclusion of non-relevant articles. Sixteen articles were excluded from pooled meta-analysis because they were case reports or case series. Three studies were excluded because they did not meet inclusion criteria. Three studies with adequate observational study design were included in pooled meta-analysis (Fig. 1).
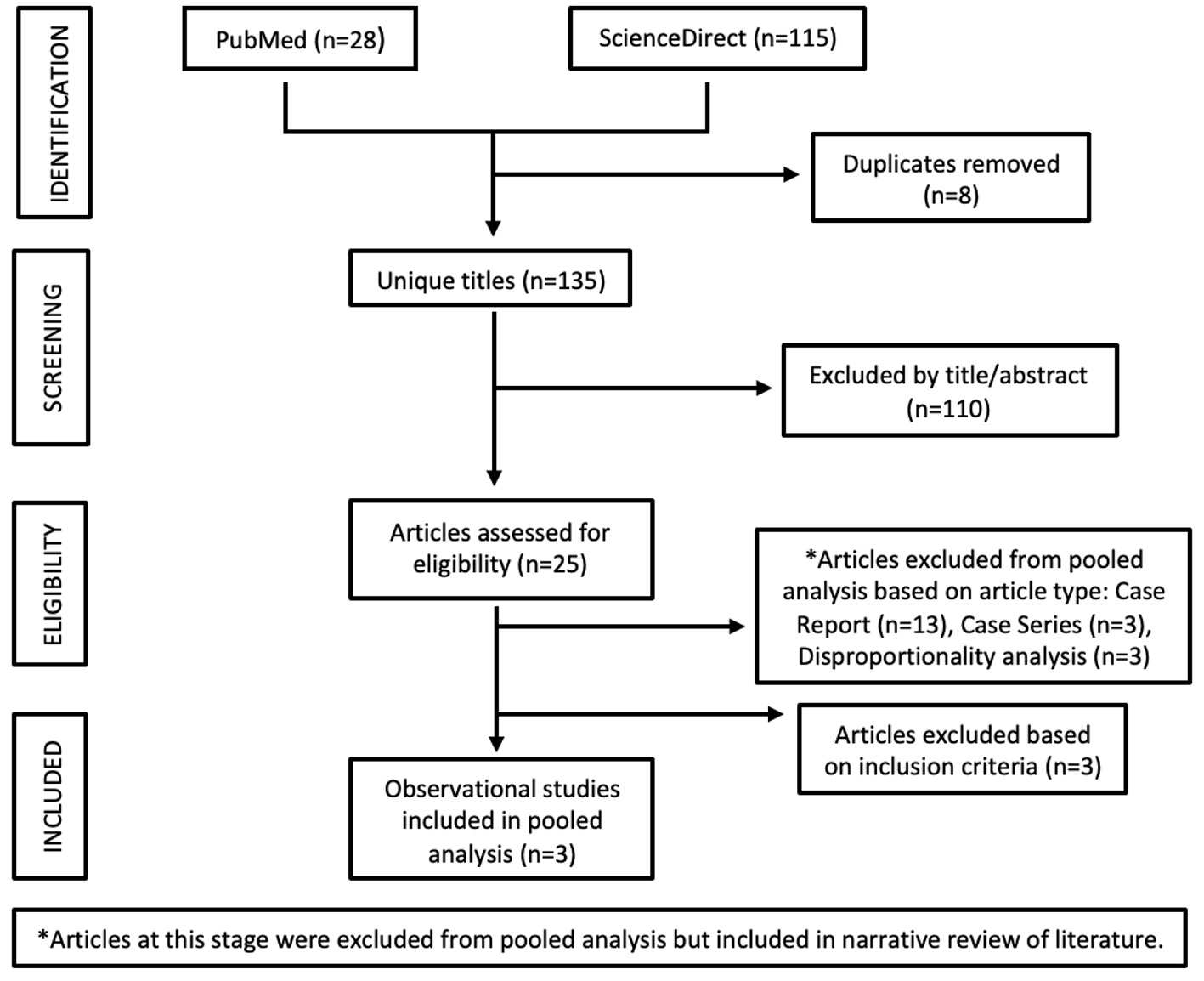 Click for large image | Figure 1. Flow diagram of study selection for pooled analysis. |
Study characteristics and quality assessment
In this meta-analysis, three studies: two retrospective cohorts and one prospective cohort observational, were identified and included in the pooled quantitative analysis (Table 1 [6-8]). The total sample of the pooled analysis was 353 individuals, including 182 patients taking RDV and 171 controls. All studies included in pooled analysis came from Italy.
 Click to view | Table 1. Characteristics of Included Studies for Pooled Meta-Analysis |
Association between RDV and bradyarrhythmia in SARS-CoV-2
The pooled analysis in Figure 2 presents the association between RDV and bradyarrhythmia in SARS-CoV-2 of the included observational studies. Independently, each included cohort study demonstrated an association between RDV administration and the development of bradyarrhythmia among SARS-CoV-2-infected patients when compared to controls. When pooled, the OR and corresponding 95% CI from the studies included was 3.27 (1.90 - 5.63). In the pooled analysis, the incidence of bradycardia in those that received RDV was 34.07% (n = 62) vs. 18.13% (n = 31) in those who did not receive RDV. Heterogeneity for this analysis was χ2 = 2.13 (P = 0.35), I2 = 6%.
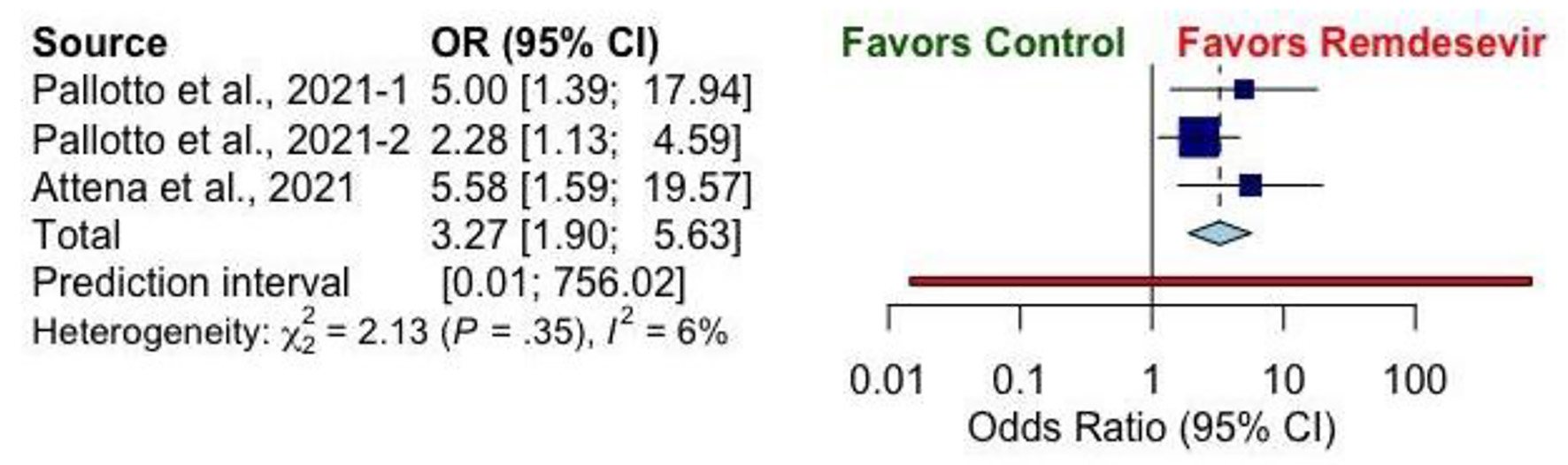 Click for large image | Figure 2. Forest plot showing risk of bradyarrhythmia among cases and controls. |
Publication bias
The funnel plot (Fig. 3) and Egger regression test demonstrated no evidence of publication bias in the pooled analysis (P = 0.2362).
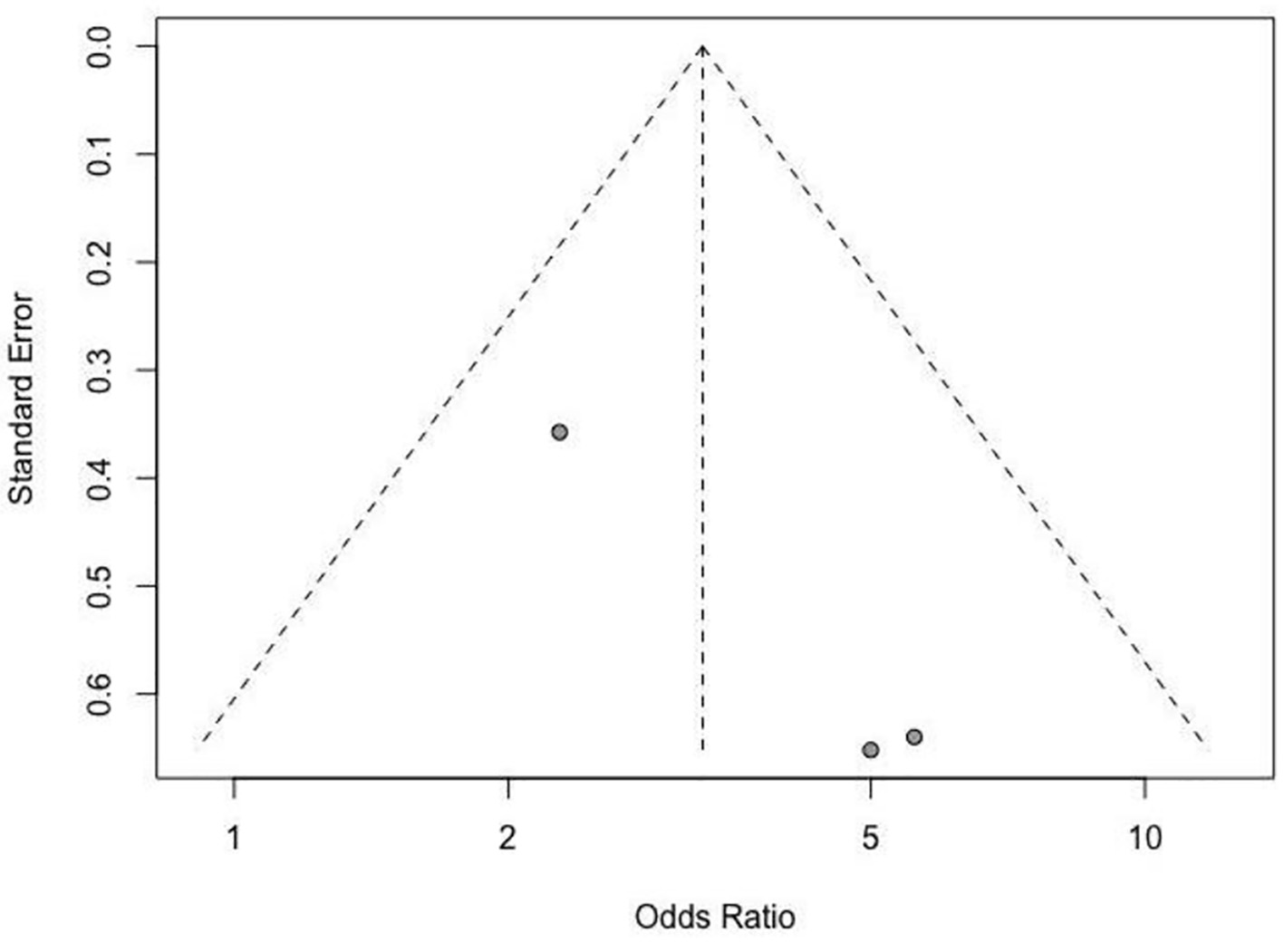 Click for large image | Figure 3. Funnel plot. |
Narrative review of literature
Only three observational cohort studies were included in the pooled analysis of the current study; however, many case reports and case series have reported bradyarrhythmia after the administration of RDV in SARS-CoV-2-infected patients. Studies reporting bradyarrhythmia after RDV therapy have been published throughout the Middle East and Western World in countries such as Iran, Qatar, Croatia, Greece, Italy, Poland, Spain, USA, and Canada (Fig. 4). Upon review of the literature, 13 case reports were identified reporting the development of bradyarrhythmia after RDV administration in COVID-19 patients [9-21]. Characteristics of these cases can be found in Table 2 [9-21]. A total of 18 individuals (ages 13 - 78 years) were identified from case presentations with an average heart rate (HR) nadir of 37.67 beats per minute (bpm). The lowest HR reported after RDV administration occurred in a 52-year-old female in Iran with a past medical history of diabetes and hypothyroidism [20]. After three doses of RDV, the patient developed angina and critical bradycardia (HR = 23 bpm), and RDV was immediately discontinued. The patient’s critical bradycardia did not respond to atropine and management was escalated to include theophylline. Four hours after discontinuation of RDV and the initiation of theophylline, the patient’s HR increased to 45 bpm, and she was discharged 2 days after admission with an HR of 55 bpm [20]. The youngest individual among case reports occurred in a 13-year-old male with a history of episodic asthma that experienced asymptomatic and non-hemodynamically significant sinus bradycardia (SB) (HR = 40 bpm) after the third dose of RDV was administered for severe bilateral pneumonia secondary to SARS-CoV-2 [18]. HR normalized in this patient within 24 h of discontinuation of RDV. Furthermore, RDV showed a temporal association with a complete atrioventricular (AV) block in a case of a 72-year-old male with a history of atrial fibrillation and lung cancer [19]. Within 24 h of RDV administration, the patient was noted to be in atrial fibrillation with ventricular rates of 30 - 40 bpm, which progressed to complete AV block on day 5 of RDV therapy and spontaneously resolution within 48 h.
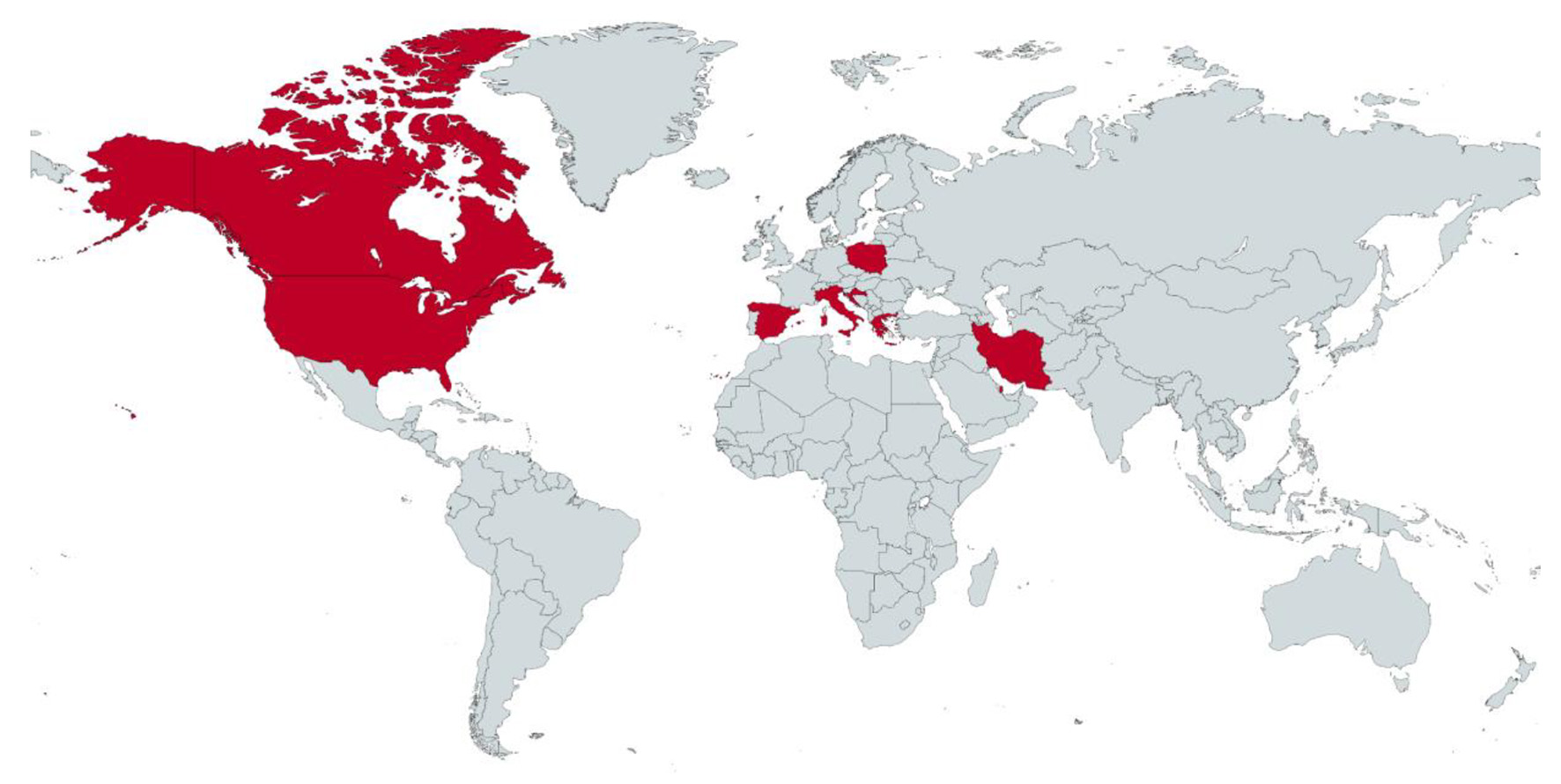 Click for large image | Figure 4. Map of countries with studies reporting bradyarrhythmia after RDV administration in SARS-CoV-2 infected patients. RDV: remdesivir; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2. |
 Click to view | Table 2. Case Presentations Reporting Bradyarrhythmia After Remdesivir Administration in SARS-CoV-2 Patients |
Three case series exploring the association between bradyarrhythmia after administration of RDV to SARS-CoV-2-infected patients were also identified in the literature [22-24]. In a case series from Poland investigating 19 hospitalized patients with marked self-limiting SB, 12 were treated with RDV before the appearance of SB (usually on day 1 or 2 of therapy) [22]. Additionally, the authors observed that SB was associated with clinical and laboratory improvement as interleukin-6 (IL-6) and C-reactive protein (CRP) were significantly lower at the onset of SB than at admission (9.3 vs. 70.0 pg/mL and 16.8 vs. 98.5 mg/L, respectively). SB was well-tolerated among this series of patients and resolved within several days. Pediatric patients have also exhibited asymptomatic SB after treatment with RDV for COVID-19. In a series of four children treated with ampicillin, RDV, and dexamethasone for COVID-19, three patients (median age 11.5 years) developed asymptomatic SB that resolved within 24 h of treatment completion or RDV discontinuation [23]. The youngest reported case of bradycardia after RDV administration was identified within this study. In a 3-month-old infant, SB was recorded after the third dose of RDV when the infant’s HR dropped to 80 bpm from 130 bpm at baseline and resolved after discontinuation of the drug [23]. To further explore the effect of RDV on arrhythmia and quantify reduction in HR, an Italian case series of 52 patients who underwent daily electrocardiogram (ECG) found a mean HR reduction of 24 ± 16 bpm, which was greater among men than women (28 ± 16 bpm vs. 18 ± 14 bpm, P < 0.05) [24]. Despite the marked reduction in HR, no arrhythmias or severe bradycardia were observed among these patients and the mean lowest HR was 61 ± 11 bpm. In fact, lower HR levels after RDV administration were associated with a less severe clinical presentation of COVID-19 by multivariate analysis (beta = 0.47, P < 0.01) [24].
Several cohort studies using real-world data have investigated the relationship of RDV and bradyarrhythmia in COVID-19 patients but did not meet the inclusion criteria for the present study. A retrospective cohort study from BronxCare Hospital Center of 37 COVID-19 patients who received RDV therapy found that 43% (n = 16) developed bradycardia [25]. The investigators found a higher incidence of bradycardia in patients who received infusion for three or more days, which is consistent with other reports. A cross-sectional pilot study investigating ECG changes 2 h after RDV infusion for 14 consecutive patients with severe COVID-19 found a statistically significant rightward deviation of the T-wave axis (42.2° prior and 69.9° after RDV administration; P = 0.047) [26]. No significant changes in heart rhythm, ventricular rate, QT interval, PR segment, QRS complex duration, P wave, QRS complex axis, rV5, or sV1 amplitudes were observed in this study (P > 0.05) but this could be attributed to the small sample size (n = 14) [26]. However, in a later retrospective study by the same authors, the frequency of bradycardia after RDV was found to be 16.8% in a cohort of 473 patients hospitalized with COVID-19 [27]. Characteristics of these observational studies and case series can be found in Table 3 [22-27].
 Click to view | Table 3. Other Studies Investigating Bradyarrhythmia After Remdesivir Administration in SARS-CoV-2 Patients |
Three separate disproportionality analyses of RDV found bradycardia among the commonly reported adverse drug reactions (ADRs) in COVID-19 patients [5, 28, 29]. RDV was the most common medication reported in the WHO Global Database of Individual Case Safety Reports (ICSRs) related to COVID-19 from January 1, 2020 to December 31, 2020; 4.6% (n = 237) of those ICSRs reported bradycardia as the ADR following RDV treatment [29]. The clinical consequences of bradycardia after RDV administration have not been well-established. However, in an analysis by Touafchia and colleagues, 80% (n = 75) of bradycardia ICSRs were serious and in 17% (n = 16) evolution of bradycardia was fatal [5].
| Discussion | ▴Top |
RDV is the only antiviral drug that has been approved by the FDA for the treatment of COVID-19 after preliminary results from the ACTT-1 study were released demonstrating that RDV reduced time to clinical recovery but had no association with a decrease in mortality. However, other randomized controlled trials (RCTs) have demonstrated different results regarding the efficacy of RDV in the treatment of COVID-19. The WHO SOLIDARITY Trial, which included 11,330 participants from 405 hospitals in 30 countries, concluded that RDV did not decrease in-hospital mortality or the need for mechanical ventilation (MV) when compared with standard of care (SOC) [30]. In fact, none of the repurposed antiviral drugs included in this trial (RDV, hydroxychloroquine, lopinavir, interferon, lopinavir + interferon) reduced mortality, hospitalization duration, or the initiation of MV. Another RCT of 857 participants, the DisCoVeRy Trial, revealed no statistically significant difference between RDV plus SOC and SOC alone (OR 0.98 (95% CI 0.77 - 1.25); P = 0.85) [31]. Moreover, when investigating the duration of treatment with RDV, there have been mixed results. In a randomized, open-label, phase 3 trial (GS-US-540-5773) of 397 participants, there was no significant difference in the efficacy between a 5-day course and a 10-day course of RDV in patients with severe COVID-19 due to SARS-CoV-2 who did not require MV at baseline [32]. In another RCT (GS-US-540-5774) that included 584 hospitalized patients with moderate COVID-19, those who received a 5-day course of RDV had better clinical status at day 11 than those who received SOC. No difference was identified in clinical status at day 11 in patients who received 10 days of RDV when compared with SOC [4]. The authors determined that these observations were of uncertain clinical importance. In the absence of alternative antiviral treatments for COVID-19, use of RDV has been widespread. Given the paucity of studies establishing RDV as superior to SOC, careful consideration should be taken when determining if RDV is a suitable treatment for a SARS-CoV-2-infected patient.
Our present study demonstrates that there is an increased risk of developing bradyarrhythmia in patients with SARS-CoV-2 infection that have been administered RDV. The composition of RDV may provide some elucidation for the observed findings. RDV is a nucleotide prodrug with antiviral activity against a broad range of viruses, including Coronaviridae, by inhibiting viral RNA-dependent RNA polymerases [4]. It is formulated with sulfobutylether B-cyclodextrin sodium for injection and has been known to penetrate deep compartments [31]. RDV is further activated to a nucleoside triphosphate analogue (GS-443902), which is an analogue of adenosine with a significantly longer half-life (11 h) [33]. Adenosine is a ubiquitous nucleoside that is released during ischemia, hypoxia, inflammation, and beta-adrenergic stimulation and its main effects on the cardiovascular system involve regulation of arterial pressure, HR, and vasomodulation [34]. The effect of adenosine on the cardiovascular system is mediated by four receptor subtypes with varying distributions in different tissue types. Notably, the A1R subtype has a high affinity for adenosine and is expressed in high levels in the atria, with greater expression in the right than the left, and activation of this receptor leads to a decrease in HR [34]. When considering the prolonged half-life of the adenosine analogue (GS-443902) after RDV is metabolized and the observed association between RDV and bradycardia, it may be wiser to treat with a 5-day regimen of RDV if used. Interestingly, a recent retrospective cohort study of 473 patients with COVID-19 that received RDV suggests a more favorable disease course in those that experience bradycardia [27]. Their analysis demonstrated that the occurrence of bradycardia on day 5 of RDV treatment was significantly associated with lower odds of death during hospitalization (OR 0.33 (95% CI 0.16 - 0.7); P = 0.014) [27]. As a result of the observed better prognosis, the authors suggest that RDV should be continued, but with increased measures of patient surveillance. This observation of a more favorable outcome among those with bradycardia was also documented in the descriptive retrospective study by Rodriguez-Guerra et al, which found that patients with bradycardia had a shorter hospitalization (15.5 vs. 20.5 days) [25].
Another important consideration when exploring the relationship between bradycardia and RDV in COVID-19 patients is the association of bradycardia and the SARS-CoV-2 infection itself. Extrapulmonary manifestations, such as those in the cardiovascular system, have been frequently reported in patients with COVID-19. Reported manifestations include acute coronary syndrome, cardiomyopathies, myocarditis, coagulopathies, and arrhythmias [35]. Exact mechanisms for the incidence of arrhythmias in the setting of COVID-19 are not fully understood, but could be the result of electrolyte abnormalities, hypoxemia, acidosis, direct pathogen effects on the sinoatrial node, or the effects of inflammatory cytokines [35, 36]. A retrospective analysis of 4,526 patients across four continents and 12 countries found that 827 patients developed an arrhythmia during infection with SARS-CoV-2 [37]. Of those who developed arrhythmia, 22.6% developed bradyarrhythmia with 12.8% bradycardia, 8.6% AV block, and 1.2% developing a pause > 3 s. Coromilas et al observed regional differences among comorbidities and the incidence in type of arrhythmia with patients in Asia having a lower burden of metabolic syndrome and tachyarrhythmia and a higher incidence of bradyarrhythmia, which the authors consider could be due to the decreased prevalence of obesity and metabolic syndrome in this population [37]. A recent case series of six cases of bradycardia attributable to COVID-19 suggests the complex systemic inflammatory response caused by SARS-CoV-2 may be the predominant factor for transient bradycardia and complete AV block observed in their study, as elevated D-dimer levels were observed in five patients [38]. The relationship of SARS-CoV and the myocardial injury has long been explored prior to the novel SARS-CoV-2. A 2009 report found SARS-CoV viral RNA in 35% of autopsied human hearts of individuals who died during the Toronto SARS outbreak: this urges consideration of direct damage to cardiomyocytes by the virus [39]. In this study, macrophage infiltration, evidence of myocardial damage, and reduced angiotensin converting enzyme 2 (ACE-2) expression were observed in the hearts of those in which SARS-CoV genome was detected. It is hypothesized that SARS-CoV downregulates the ACE-2 system and can mediate myocardial inflammation and damage [39]. Relative bradycardia, an inappropriately low HR in response to increased body temperature, has also become an area of interest in patients suffering from COVID-19. Capoferri et al retrospectively reviewed the electronic medical records of patients with confirmed SARS-CoV-2 infection and found 40 patients to have relative bradycardia (36% of all COVID-19 patients and 56% of febrile COVID-19 patients) [36]. Hu and colleagues found that a third of the patients with severe illness (n = 8) developed SB with HRs ranging from 45 to 58 bpm, and that SB most often occurred during sleep [40]. The authors hypothesize that an important risk factor for SB in COVID-19 patients may be deep sleep or sedation. The growing evidence of bradyarrhythmia and COVID-19 needs to be further explored, both in the setting of RDV treatment and SOC.
The present study has several limitations that should be considered. First, only three observational studies were identified that investigated the relationship between bradyarrhythmia and RDV in patients with SARS-CoV-2 with adequate study design. The small number of studies that could be meaningfully pooled is an important limitation to consider. Given the breadth of cases that have been reported in the literature and the biochemical makeup of RDV, more studies are needed to compare the development of bradyarrhythmia after RDV administration in SARS-CoV-2-infected patients. Second, all three of the observational studies that met the inclusion criteria came from the same country, Italy. More studies are needed in Western and Eastern populations to investigate this relationship and extrapolate the findings suggested in the present study. With data from Coromilas et al suggesting a higher burden of bradyarrhythmia among Asian cohorts with COVID-19, it will be important to explore to what extent, if any, RDV has on this observation. Additionally, we should appreciate that the treatment and sequelae of SARS-CoV-2 infection are rapidly evolving and there is a scarcity of studies that have investigated niche sequelae such as bradyarrhythmia following RDV administration for the treatment of this novel virus.
Conclusion
Our meta-analysis of available real-world data suggests there is an increased risk of developing bradycardia in patients with SARS-CoV-2 that are treated with RDV. Although, the importance of this observation is of uncertain clinical significance as some observational studies have reported more favorable outcomes among patients who experience bradycardia after RDV therapy. Clinicians treating patients with SARS-CoV-2 infection should remain vigilant when treating with RDV given the breadth of observational studies reporting bradycardia upon administration. Future investigations using well-designed cohort studies are needed to further explore this relationship, as the knowledge of SARS-CoV-2 infection and its treatment are rapidly evolving.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Not applicable.
Author Contributions
Safa Al-Jammali contributed to study design, manuscript development, and editing; Rana Al-Zakhari contributed to manuscript development and editing; Nicholas Sheets contributed to study design, study selection, data collection, quality assessment, manuscript development, and figure and table design and development; Arun Mahtani contributed to statistical analysis, quality assessment, manuscript development, and figure design and development; Veronika Stefanishina contributed to study selection and table development; Nidal Isber contributed to oversight and editing of final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; COVID-19: coronavirus disease 2019; RDV: remdesivir; EUA: Emergency Use Authorization; CI: confidence interval; OR: odds ratio; FDA: US Food and Drug Administration; HR: heart rate; AV: atrioventricular; IL-6: interleukin-6; CRP: C-reactive protein; bpm: beats per minute; AF: atrial fibrillation; SB: sinus bradycardia; SOC: standard of care; ACE-2: angiotensin converting enzyme 2; MV: mechanical ventilation; WHO: World Health Organization; ECG: electrocardiogram; ADR: adverse drug reaction; ICSR: Individual Case Safety Reports; ACTT-1: Adaptive COVID-19 Treatment Trial
| References | ▴Top |
- Li H, Liu SM, Yu XH, Tang SL, Tang CK. Coronavirus disease 2019 (COVID-19): current status and future perspectives. Int J Antimicrob Agents. 2020;55(5):105951.
doi pubmed - Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395(10224):565-574.
doi - Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, Hohmann E, et al. Remdesivir for the treatment of COVID-19 - final report. N Engl J Med. 2020;383(19):1813-1826.
doi pubmed - Spinner CD, Gottlieb RL, Criner GJ, Arribas Lopez JR, Cattelan AM, Soriano Viladomiu A, Ogbuagu O, et al. Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial. JAMA. 2020;324(11):1048-1057.
doi pubmed - Touafchia A, Bagheri H, Carrie D, Durrieu G, Sommet A, Chouchana L, Montastruc F. Serious bradycardia and remdesivir for coronavirus 2019 (COVID-19): a new safety concerns. Clin Microbiol Infect. 2021;27(5):791.E5-791.E8.
doi pubmed - Pallotto C, Suardi LR, Gabbuti A, Esperti S, Mecocci L, Blanc P. Potential remdesivir-related transient bradycardia in patients with coronavirus disease 2019 (COVID-19). J Med Virol. 2021;93(5):2631-2634.
doi pubmed - Pallotto C, Blanc P, Esperti S, Suardi LR, Gabbuti A, Vichi F, Mecocci L, et al. Remdesivir treatment and transient bradycardia in patients with coronavirus diseases 2019 (COVID-19). J Infect. 2021;83(2):237-279.
doi pubmed - Attena E, Albani S, Maraolo AE, Mollica M, De Rosa A, Pisapia R, Fiorentino G, et al. Remdesivir-induced bradycardia in COVID-19: a single center prospective study. Circ Arrhythm Electrophysiol. 2021;14(7):e009811.
- Abdelmajid A, Osman W, Musa H, Elhiday H, Munir W, Al Maslamani MA, Elmekaty EZ. Remdesivir therapy causing bradycardia in COVID-19 patients: Two case reports. IDCases. 2021;26:e01254.
doi pubmed - Barkas F, Styla CP, Bechlioulis A, Milionis H, Liberopoulos E. Sinus bradycardia associated with remdesivir treatment in COVID-19: a case report and literature review. J Cardiovasc Dev Dis. 2021;8(2):18.
doi pubmed - Ching PR, Lee C. Remdesivir-associated bradycardia. BMJ Case Rep. 2021;14(9);e245289.
- Chow EJ, Maust B, Kazmier KM, Stokes C. Sinus bradycardia in a pediatric patient treated with remdesivir for acute coronavirus disease 2019: a case report and a review of the literature. J Pediatric Infect Dis Soc. 2021;10(9):926-929.
doi pubmed - Day LB, Abdel-Qadir H, Fralick M. Bradycardia associated with remdesivir therapy for COVID-19 in a 59-year-old man. CMAJ. 2021;193(17):E612-E615.
doi pubmed - Gubitosa JC, Kakar P, Gerula C, Nossa H, Finkel D, Wong K, Khatri M, et al. Marked sinus bradycardia associated with remdesivir in COVID-19: a case and literature review. JACC Case Rep. 2020;2(14):2260-2264.
doi pubmed - Gupta AK, Parker BM, Priyadarshi V, Parker J. Cardiac adverse events with remdesivir in COVID-19 infection. Cureus. 2020;12(10):e11132.
doi - Jacinto JP, Patel M, Goh J, Yamamura K. Remdesivir-induced symptomatic bradycardia in the treatment of COVID-19 disease. HeartRhythm Case Rep. 2021;7(8):514-517.
doi pubmed - Maheshwari M, Athiraman H. Bradycardia related to remdesivir during COVID-19: persistent or permanent? Cureus. 2021;13(11):e19919.
doi - Sanchez-Codez MI, Rodriguez-Gonzalez M, Gutierrez-Rosa I. Severe sinus bradycardia associated with Remdesivir in a child with severe SARS-CoV-2 infection. Eur J Pediatr. 2021;180(5):1627.
doi pubmed - Selvaraj V, Bavishi C, Patel S, Dapaah-Afriyie K. Complete heart block associated with Remdesivir in COVID-19: a case report. Eur Heart J Case Rep. 2021;5(7):ytab200.
- Shirvani M, Sayad B, Shojaei L, Amini A, Shahbazi F. Remdesivir-associated significant bradycardia: a report of three cases. J Tehran Heart Cent. 2021;16(2):79-83.
doi pubmed - Sneij E, Kohli V, Al-Adwan SA, Mealor A. Remdesivir causing profound bradycardia. J Am Coll Cardiol. 2021;77(18):2037.
doi - Elikowski W, Fertala N, Zawodna-Marszalek M, Rajewska-Tabor J, Swidurski W, Wajdlich D, Marszalek A, et al. Marked self-limiting sinus bradycardia in COVID-19 patients not requiring therapy in the intensive care unit - case series report. Pol Merkur Lekarski. 2021;49(292):295-302.
- Eleftheriou I, Liaska M, Krepis P, Dasoula F, Dimopoulou D, Spyridis N, Tsolia M. Sinus Bradycardia in children treated with remdesivir for COVID-19. Pediatr Infect Dis J. 2021;40(9):e356.
doi pubmed - Brunetti ND, Poliseno M, Bottalico IF, Centola A, Montemurro L, Sica S, Santantonio T, et al. Safety and heart rate changes in COVID-19 patients treated with remdesivir. Int J Infect Dis. 2021;112:254-257.
doi pubmed - Rodriguez-Guerra M, Mendoza C, Kandhi S, Sun H, Saad M, Vittorio T. Cardiac arrhythmia related to remdesivir in COVID-19. ISMMS Journal of Science and Medicine. 2021;1(2):15.
doi - Bistrovic P, Lucijanic M. Remdesivir might induce changes in electrocardiogram beyond bradycardia in patients with coronavirus disease 2019-The pilot study. J Med Virol. 2021;93(10):5724-5725.
doi pubmed - Bistrovic P, Manola S, Lucijanic M. Bradycardia during remdesivir treatment might be associated with improved survival in patients with COVID-19: a retrospective cohort study on 473 patients from a tertiary centre. Postgrad Med J. 2021.
- Singh A, Kamath A. Assessment of adverse events associated with remdesivir use for coronavirus disease 2019 using real-world data. Expert Opin Drug Saf. 2021;20(12):1559-1564.
doi pubmed - Rocca E, Gauffin O, Savage R, Vidlin SH, Grundmark B. Remdesivir in the COVID-19 pandemic: an analysis of spontaneous reports in VigiBase during 2020. Drug Saf. 2021;44(9):987-998.
doi pubmed - WHO Solidarity Trial Consortium, Pan H, Peto R, Henao-Restrepo AM, Preziosi MP, Sathiyamoorthy V, Abdool Karim Q, et al. Repurposed antiviral drugs for COVID-19 - interim WHO solidarity trial results. N Engl J Med. 2021;384(6):497-511.
doi pubmed - Ader F, Bouscambert-Duchamp M, Hites M, Peiffer-Smadja N, Poissy J, Belhadi D, Diallo A, et al. Remdesivir plus standard of care versus standard of care alone for the treatment of patients admitted to hospital with COVID-19 (DisCoVeRy): a phase 3, randomised, controlled, open-label trial. Lancet Infect Dis. 2022;22(2):209-221.
doi - Goldman JD, Lye DCB, Hui DS, Marks KM, Bruno R, Montejano R, Spinner CD, et al. Remdesivir for 5 or 10 days in patients with severe COVID-19. N Engl J Med. 2020;383(19):1827-1837.
doi pubmed - Nabati M, Parsaee H. Potential cardiotoxic effects of remdesivir on cardiovascular system: a literature review. Cardiovasc Toxicol. 2022;22(3):268-272.
doi pubmed - Guieu R, Deharo JC, Maille B, Crotti L, Torresani E, Brignole M, Parati G. Adenosine and the cardiovascular system: the good and the bad. J Clin Med. 2020;9(5):1366.
doi pubmed - Douedi S, Mararenko A, Alshami A, Al-Azzawi M, Ajam F, Patel S, Douedi H, et al. COVID-19 induced bradyarrhythmia and relative bradycardia: An overview. J Arrhythm. 2021;37(4):888-892.
doi pubmed - Capoferri G, Osthoff M, Egli A, Stoeckle M, Bassetti S. Relative bradycardia in patients with COVID-19. Clin Microbiol Infect. 2021;27(2):295-296.
doi pubmed - Coromilas EJ, Kochav S, Goldenthal I, Biviano A, Garan H, Goldbarg S, Kim JH, et al. Worldwide survey of COVID-19-associated arrhythmias. Circ Arrhythm Electrophysiol. 2021;14(3):e009458.
- Amir M, Yoseph H, Farisi ATA, Phie JKP, Adam ATS. Symptomatic bradycardia in COVID-19 hospitalized patients: a case series. Int J Infect Dis. 2021;111:1-4.
doi pubmed - Oudit GY, Kassiri Z, Jiang C, Liu PP, Poutanen SM, Penninger JM, Butany J. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest. 2009;39(7):618-625.
doi pubmed - Hu L, Gong L, Jiang Z, Wang Q, Zou Y, Zhu L. Clinical analysis of sinus bradycardia in patients with severe COVID-19 pneumonia. Crit Care. 2020;24(1):257.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


