| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 14, Number 1, February 2023, pages 32-37
Assessing Correlation Between Thoracic Impedance and Remotely Monitored Pulmonary Artery Pressure in Chronic Systolic Heart Failure
Ankita Aggarwala, c, Zubair Khanb, Christian Machadoa, Marcel Zughaiba
aDepartment of Cardiology Ascension Providence Hospital, Southfield, MI, USA
bCardiovascular Services, North Alabama Medical Center, Florence, AL 35630, USA
cCorresponding Author: Ankita Aggarwal, Department of Cardiology, Ascension Providence Hospital, Southfield, MI, USA
Manuscript submitted November 8, 2022, accepted January 6, 2023, published online February 25, 2023
Short title: TI and PAP in Heart Failure
doi: https://doi.org/10.14740/cr1447
| Abstract | ▴Top |
Background: Heart failure (HF) readmission continues to be a major health problem. Monitoring pulmonary artery pressure (PAP) and thoracic impedance (TI) are the two modalities utilized for early identification of decompensation in HF patients. We aimed to assess the correlation between these two modalities in patients who simultaneously had both the devices.
Methods: Patients with history of New York Heart Association class III systolic HF with a pre-implanted intracardiac defibrillator (ICD) capable of monitoring TI and pre-implanted CardioMEMs™ remote HF monitoring device were included. Hemodynamic data including TI and PAPs were measured at baseline and then weekly. Weekly percentage change was then calculated as: Weekly percentage change = (week 2 - week1)/week 1 × 100. Variability between the methods was expressed by Bland-Altman analysis. Significance was determined as a P-value < 0.05.
Results: Nine patients met the inclusion criteria. There was no significant correlation between the assessed weekly percentage changes in pulmonary artery diastolic pressure (PAdP) and TI measurements (r = -0.180, P = 0.065). Using Bland-Altman analytic methods, both methods had no significant difference in agreement (0.011±0.094%, P = 0.215). With the linear regression model applied for Bland-Altman analysis, the two methods appeared to have proportional bias without agreement (unstandardized beta-coefficient of 1.91, t 22.9, P ≤ 0.001).
Conclusion: Our study demonstrated that variations exist between measurement of PAdP and TI; however, there is no significant correlation between weekly variations between them.
Keywords: Pulmonary artery pressures; Thoracic impedance; Congestive heart failure
| Introduction | ▴Top |
Heart failure (HF) is a major health problem with a continuous rise in its prevalence despite major advances in pharmacotherapy and adjunctive therapies over the past few decades [1]. It affects approximately 6 million or 1.8% of the total US population as per the 2021 American Heart Association Statistical Update [2]. HF is one of major causes of hospitalization [3] and is associated with high readmission rates and thus increased mortality [4]. Over the past several decades, multiple attempts have been made to improve HF readmission rates including the introduction of new classes of medications. In the Paradigm-HF trial [5], the use of angiotensin receptor-neprilysin inhibitor (ARNI) resulted in a decreased rate of readmission for HF. More recently, sodium-glucose co-transporter-2 (SGLT-2) inhibitors dapagliflozin [6] and empagliflozin [7, 8] also similarly reduced HF hospitalization when added to goal-directed medical therapy. Despite these significant recent medical advances, readmissions in patients with chronic HF have continued to rise over the past decade [9]. About 31% of the patients are readmitted within 90 days [9].
The most common reason for admission with acute HF is congestion [10]. Continued efforts are being made to utilize remote monitoring to identify patients at early stages of congestion and institute therapeutic changes that reduce readmission. Remote monitoring of pulmonary artery pressure (PAP) using the CardioMEMS™ HF System (Abbott Laboratories, Sylmar, CA) has shown promise in reducing readmission rate and thus improving outcomes. However, randomized controlled trials have shown conflicting results. The COMPASS-HF [11] trial showed that assessing implantable continuous hemodynamic monitor-guided care did not significantly reduce total HF-related events compared with optimal medical management. On the other hand, the CHAMPION trial, a major randomized controlled trial, showed hemodynamic monitoring using PAP led to increased medication changes and reduced HF hospitalization [12]. A more recent trial, the GUIDE-HF [13], failed to show a benefit of hemodynamic monitoring on the composite endpoint rate of mortality and total HF events. However, the results were affected by a COVID-19 pandemic mainly driven by significant reduction in HF hospitalization in the control group during the pandemic. This was possibly related to multiple factors including but not limited to changes in patient compliance with medications and dietary restrictions, access to health care and possible effect of COVID-19 on disease progression. However, an analysis of the pre-pandemic period showed reduced HF hospitalization rate in the management group compared with the control group. Despite the proven benefit, its large-scale adoption in the real world continues to lag behind [14].
Another commonly utilized non-invasive modality is measuring the thoracic impedance (TI) using the cardiovascular implantable electronic devices (CIED). Studies have shown that monitoring of TI is correlated to increase in pulmonary capillary wedge pressure and congestion [15]. It was also found that decreased TI was independently associated with increased risk of HF hospitalization [16, 17]. As a result, algorithms were developed and studied to assess utility of this modality in predicting acute decompensation. However, these studies showed low sensitivity and high inter-patient variability in predicting episodes of acute decompensation in patients with chronic HF [18]. In the DOT-HF study, a randomized controlled trial management based on alerts from decreased TI failed to decrease hospitalization and/or improve mortality when compared to usual care [19].
In summary, while the evidence has shown potential benefit of monitoring PAP, the utility of TI remains uncertain. We designed this prospective study with an aim to assess the correlation between these two modalities in patients who simultaneously had both the devices to fully understand the clinical usefulness of these devices in patients with chronic HF.
| Materials and Methods | ▴Top |
Study design
Our research protocol was developed and approved by the institutional review board (IRB). The study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration. We included patients with history of New York Heart Association (NYHA) class III systolic HF longer than 6 months with a pre-implanted intracardiac defibrillator (ICD) capable of monitoring TI (St. Jude Medical’s Corvue™ Birmingham, MN) and pre-implanted CardioMEMs™ remote HF monitoring device. Eligible patients were identified using the Merlin.net™ database of patients with CardioMEMs™ device previously implanted at our institution (Ascension Providence Hospital, Southfield) and cross-checking them with our pacemaker clinic to assess whether they have implanted ICDs which can measure TI (St. Jude Medical’s Corvue™). After initial identification, subjects were brought in for a routine clinic visit where consent for enrolment in the study was obtained. During this initial visit, each patient was assessed for volume status and current medications, baseline assessment of pulmonary artery diastolic pressure (PAdP) via CardioMems device along with their St. Jude ICD interrogation. Patients were subsequently prospectively followed over a period of 12 weeks. Medication changes, if needed, were left to the treating cardiologist’s discretion.
Remote hemodynamic data were acquired from the St. Jude portal (Merlin) for weekly CardioMems PAdP transmissions and biweekly transmissions from CorVue TI recordings. Weekly PAdP values (mm Hg) were averaged (defined to be valid for at least three valid transmissions per week) for an individual subject during the follow-up duration.
TI data were acquired from St. Jude’s CorVue remote device monitoring system. The weekly values (ohms) were acquired from reconstructed graphs/curves using the web-based software program, WebPlotDigitizer [20]. For comparison across a uniform variable for the two methods, weekly PAdP measurements were averaged. Weekly percentage change was then calculated as: Weekly percentage change = (week 2 - week1)/week 1 × 100.
Statistical analysis
All statistical analyses were performed using IBM SPSS, version 21.0 [21]. Summary statistics are presented as N (%) for categorical variables; continuous variables are presented as mean ± standard deviation (SD). With presence of outliers and skewed distribution of TI data (P = 0.161 for weekly change in PAdP and P = 0.015 for weekly change in TI by Shapiro-Wilk test), non-parametric, Spearman correlation coefficient was utilized for the comparison of weekly percentage change in PAPs obtained with CardioMEMS™ and CorVue TI data. Variability between the methods was expressed relative to the average PAdP plus 2 SDs by Bland-Altman analysis [22]. Significance was determined as a P-value < 0.05.
| Results | ▴Top |
We identified nine patients in our practice with previously implanted St. Jude ICDs as well as CardioMems devices. Baseline demographic, clinical data, pharmacologic regimen and baseline hemodynamic PAP readings from CardioMems are presented in Table 1.
 Click to view | Table 1. Baseline Characteristics of the Included Patients |
Aside from one subject (with missing CardioMems transmissions for weeks 3 and 12), all subjects had at least three reliable PAdP transmissions with a total of 106 readings included in the final analysis. Linear regression and agreement plots for weekly percentage changes in PAdP from CardioMEMS™ and CorVue TI measurements are shown in Figure 1. There was no significant correlation between the assessed weekly percentage changes in PAdP and TI measurements (r = -0.180, P = 0.065). Using Bland-Altman analytic methods, both methods had no significant difference in agreement (0.011±0.094%, P = 0.215). With the linear regression model applied for Bland-Altman analysis, the two methods appeared to have proportional bias without agreement (unstandardized beta coefficient of 1.91, t 22.9, P ≤ 0.001) (Fig. 2).
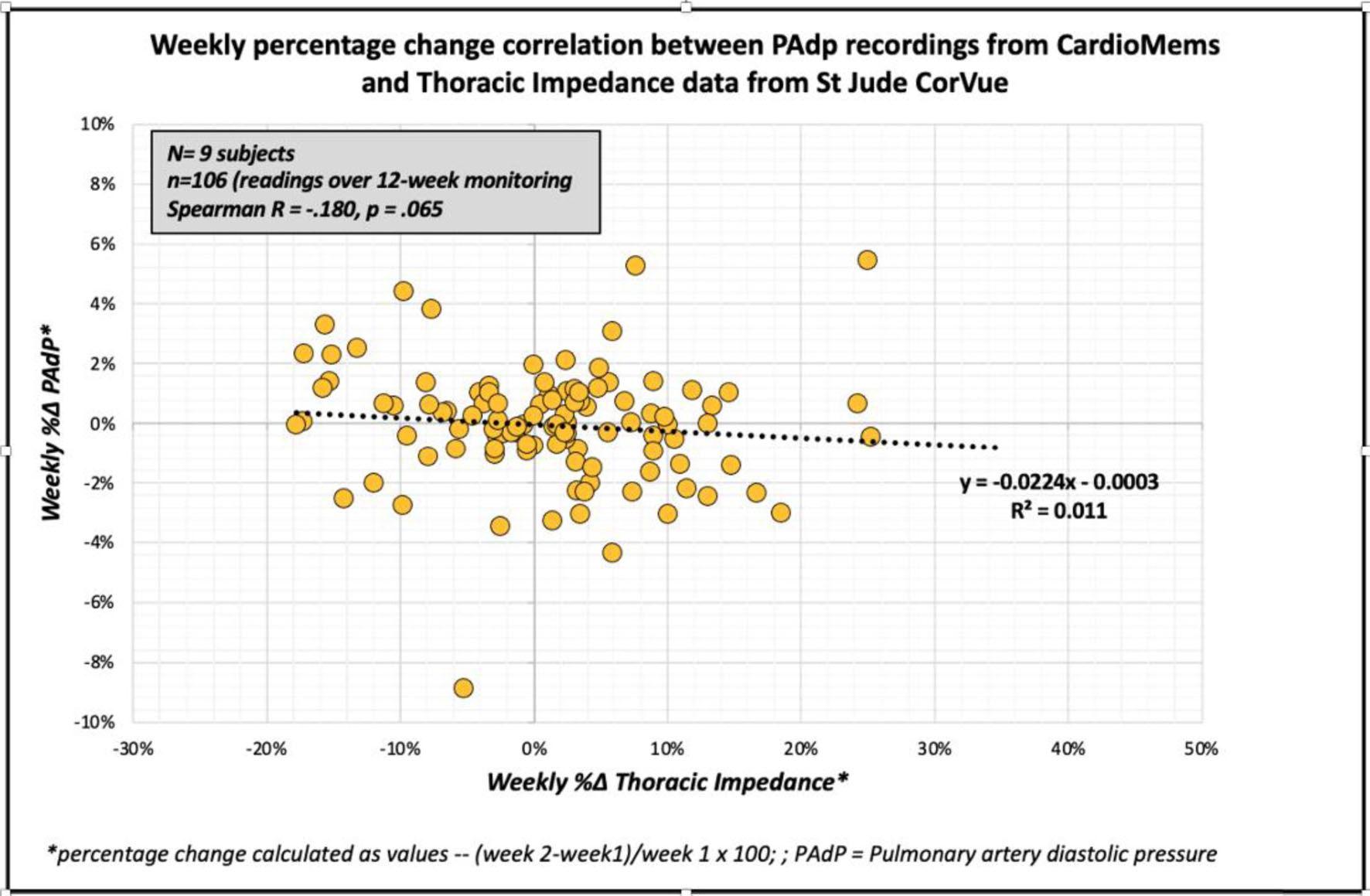 Click for large image | Figure 1. Correlation between weekly percentage changes in pulmonary artery pressure and thoracic impedance. |
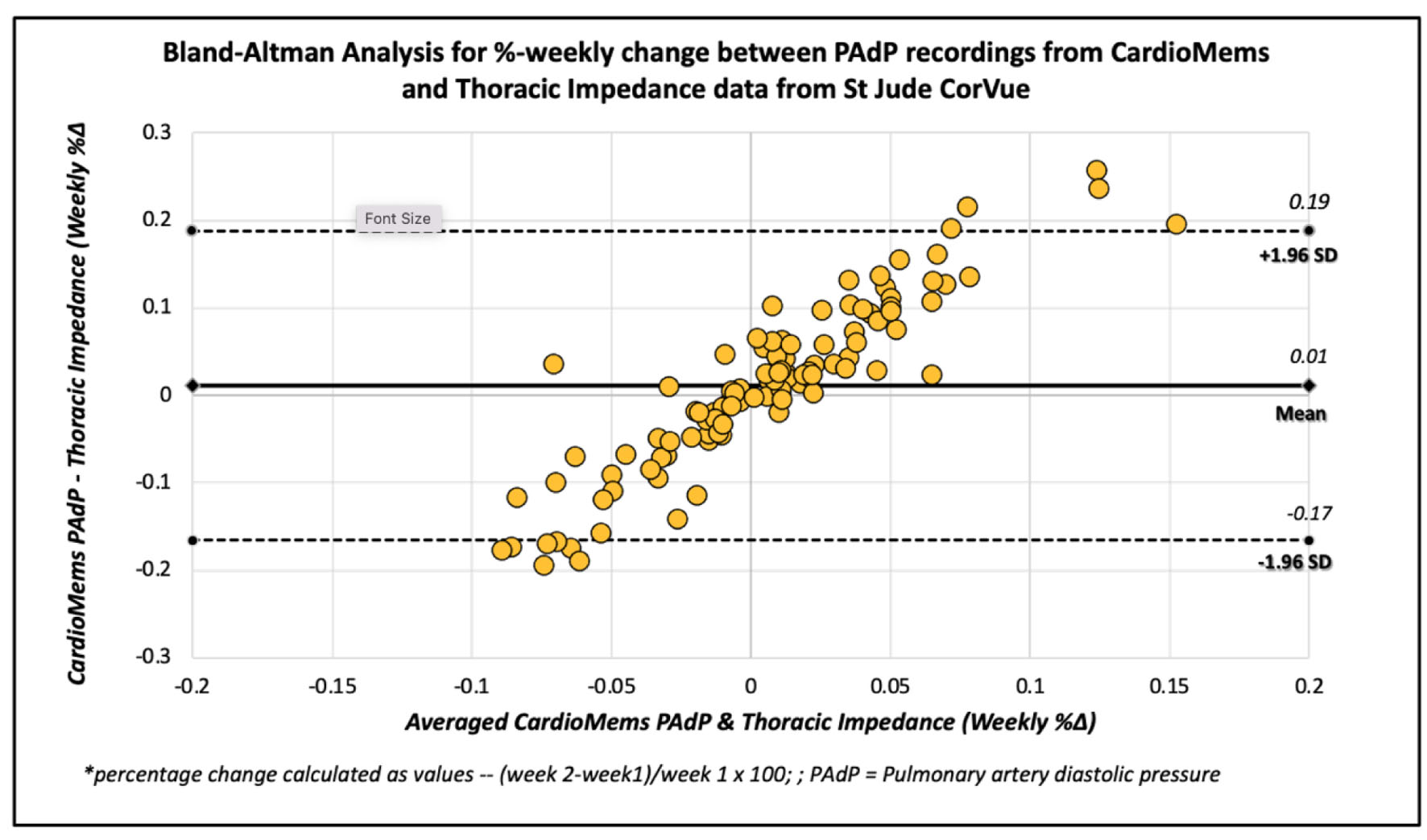 Click for large image | Figure 2. Bland-Altman analysis of weekly percentage changes in pulmonary artery pressure and thoracic impedance. |
| Discussion | ▴Top |
Reducing readmission among patients with HF remains an unmet need. Recent advances have focused on identifying patients earlier in the clinical congestive cascade prior to development of symptoms [23]. As per the congestive cascade, the increase in filling pressure significantly precedes decrease in TI and occurs about 20 and 10 days prior to the development of symptoms, respectively [24].
Theoretically, accumulation of intrathoracic fluid during pulmonary congestion results in better conductance, causing a corresponding decrease in impedance. Thus, a decrease in TI should allow prediction of congestion leading to subsequent HF readmission. This hypothesis was described in an early study by Yu et al [15]. They found that TI decreased before each HF hospitalization and was associated with increased pulmonary capillary wedge pressure during hospitalization. However, management based on these changes failed to decrease readmission rates in randomized controlled trials [19]. One of the explanations is that there are numerous factors contributing to variations in measurements of TI by implanted devices. Tang and colleagues described that electrode placements, blood content, viscosity, skin moisture, body composition, body habitus, aortic valve disorders, volume status of a patient, as well as environmental noise among others can contribute to changes in TI values [25]. In addition to that, the short period between occurrence of changes in TI and clinical decongestion does not allow enough time for clinical decision-making including medications changes to affect outcomes.
This stands in contrast to remote monitoring of PAP. As the increase in filling pressure precedes the decrease in impedance by approximately 10 days, it is presumed to be more beneficial than measuring TI and will allow for earlier identification of patients with decompensation. Multiple studies have assessed the utility of these two modalities in reducing HF readmission and thus improving outcomes independently. We aimed to assess the simultaneous correlation between the two modalities. Our study demonstrated that variations exist between measurement of PAdP and TI; however, there is no significant correlation between weekly variations between them. A similar study by Volodarskiy et al found no significant correlation between changes in PAP and TI during follow-up period in chronic HF patients [26]. Furthermore, they found that no significant variations existed in either of the two modalities prior to admission with decompensation [26].
Another study that assessed correlation between the two modalities was conducted by Perego et al [27]. They found that there was a significant negative correlation in variation of PAP and TI from baseline value. However, like our study results, the weekly variations in these two parameters did not correlate.
Putting all the data from clinical trial and our study in perspective, it may be reasonable to conclude that hemodynamic monitoring using PAP is more beneficial than monitoring of TI as it allows for earlier identification of possibly decompensating patients and thus leads to improved outcomes.
Limitations
This is a single-center study and carries inherent limitations. Besides a small sample size, local patient demographics and characteristics could further limit the generalization of these findings. While we used only a single device algorithm (St. Jude CorVue) to assess TI, this cannot be generalized to other device algorithms like Medtronic’s OptiVol or BioTronik. While the weekly percentage variation appears to correlate, with limited sample size and short duration of follow-up with no HF-related readmission during the study duration, it is not possible to make any conclusions on the preceding variations in PAdP or TI. Larger randomized trials are needed to predict which variables are better predictors of HF-related admissions.
Conclusion
Significant PAP and TI variations can exist in patients with HF. Our study showed there was no significant correlation when assessing weekly variations between TI and PAP values. Although TI values have shown negative correlation with PAP measurement, data are lacking regarding their clinical benefit in capturing patients in the initial stages of the HF cascade and reducing hospitalizations related to acute exacerbations. PAP remote monitoring may be a more useful tool to monitor HF patients and to reduce their hospital readmissions for decompensation.
Acknowledgments
None to declare.
Financial Disclosure
There was no specific funding source to be mentioned.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
All subjects provided written informed consent.
Author Contributions
ZK designed and performed the study. AA and MZ drafted the manuscript and did critical editing. AA and ZK assisted and supported in sample collection and subsequent analysis with statistics. MZ and CM carefully supervised this manuscript preparation and writing.
Data Availability
The authors declare that data supporting the findings of this study are available within the article
Abbreviations
ICD: intracardiac defibrillator; NYHA: New York Heart Association; PAdP: pulmonary artery diastolic pressure; TI: thoracic impedance; PAP: pulmonary artery pressure; CIED: cardiovascular implantable electronic devices
| References | ▴Top |
- Heidenreich PA, Albert NM, Allen LA, Bluemke DA, Butler J, Fonarow GC, Ikonomidis JS, et al. Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail. 2013;6(3):606-619.
doi pubmed - Roger VL. Epidemiology of heart failure: a contemporary perspective. Circ Res. 2021;128(10):1421-1434.
doi pubmed - Jackson SL, Tong X, King RJ, Loustalot F, Hong Y, Ritchey MD. National Burden of Heart Failure Events in the United States, 2006 to 2014. Circ Heart Fail. 2018;11(12):e004873.
doi pubmed - Lan T, Liao YH, Zhang J, Yang ZP, Xu GS, Zhu L, Fan DM. Mortality and readmission rates after heart failure: a systematic review and meta-analysis. Ther Clin Risk Manag. 2021;17:1307-1320.
doi pubmed - McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, Rouleau JL, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993-1004.
doi pubmed - McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, Ponikowski P, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995-2008.
doi pubmed - Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, Januzzi J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413-1424.
doi pubmed - Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Bohm M, Brunner-La Rocca HP, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451-1461.
doi pubmed - Khan MS, Sreenivasan J, Lateef N, Abougergi MS, Greene SJ, Ahmad T, Anker SD, et al. Trends in 30- and 90-day readmission rates for heart failure. Circ Heart Fail. 2021;14(4):e008335.
doi pubmed - Boorsma EM, Ter Maaten JM, Damman K, Dinh W, Gustafsson F, Goldsmith S, Burkhoff D, et al. Congestion in heart failure: a contemporary look at physiology, diagnosis and treatment. Nat Rev Cardiol. 2020;17(10):641-655.
doi pubmed - Bourge RC, Abraham WT, Adamson PB, Aaron MF, Aranda JM, Jr., Magalski A, Zile MR, et al. Randomized controlled trial of an implantable continuous hemodynamic monitor in patients with advanced heart failure: the COMPASS-HF study. J Am Coll Cardiol. 2008;51(11):1073-1079.
doi pubmed - Givertz MM, Stevenson LW, Costanzo MR, Bourge RC, Bauman JG, Ginn G, Abraham WT, et al. Pulmonary Artery Pressure-Guided Management of Patients With Heart Failure and Reduced Ejection Fraction. J Am Coll Cardiol. 2017;70(15):1875-1886.
doi pubmed - Lindenfeld J, Zile MR, Desai AS, Bhatt K, Ducharme A, Horstmanshof D, Krim SR, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. Lancet. 2021;398(10304):991-1001.
doi pubmed - Hernandez GA, Lemor A, Blumer V, Rueda CA, Zalawadiya S, Stevenson LW, Lindenfeld J. Trends in utilization and outcomes of pulmonary artery catheterization in heart failure with and without cardiogenic shock. J Card Fail. 2019;25(5):364-371.
doi pubmed - Yu CM, Wang L, Chau E, Chan RH, Kong SL, Tang MO, Christensen J, et al. Intrathoracic impedance monitoring in patients with heart failure: correlation with fluid status and feasibility of early warning preceding hospitalization. Circulation. 2005;112(6):841-848.
doi pubmed - Catanzariti D, Lunati M, Landolina M, Zanotto G, Lonardi G, Iacopino S, Oliva F, et al. Monitoring intrathoracic impedance with an implantable defibrillator reduces hospitalizations in patients with heart failure. Pacing Clin Electrophysiol. 2009;32(3):363-370.
doi pubmed - Perego GB, Landolina M, Vergara G, Lunati M, Zanotto G, Pappone A, Lonardi G, et al. Implantable CRT device diagnostics identify patients with increased risk for heart failure hospitalization. J Interv Card Electrophysiol. 2008;23(3):235-242.
doi pubmed - Maier SKG, Paule S, Jung W, Koller M, Ventura R, Quesada A, Bordachar P, et al. Evaluation of thoracic impedance trends for implant-based remote monitoring in heart failure patients - Results from the (J-)HomeCARE-II Study. J Electrocardiol. 2019;53:100-108.
doi pubmed - van Veldhuisen DJ, Braunschweig F, Conraads V, Ford I, Cowie MR, Jondeau G, Kautzner J, et al. Intrathoracic impedance monitoring, audible patient alerts, and outcome in patients with heart failure. Circulation. 2011;124(16):1719-1726.
doi pubmed - Rohatgi A. WebPlotDigitizer n.d. https://automeris.io/WebPlotDigitizer.
- IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.
- Giavarina D. Understanding Bland Altman analysis. Biochem Med (Zagreb). 2015;25(2):141-151.
doi pubmed - Adamson PB. Pathophysiology of the transition from chronic compensated and acute decompensated heart failure: new insights from continuous monitoring devices. Curr Heart Fail Rep. 2009;6(4):287-292.
doi pubmed - Arrigo M, Nijst P, Rudiger A. Optimising heart failure therapies in the acute setting. Card Fail Rev. 2018;4(1):38-42.
doi pubmed - Tang WH, Tong W. Measuring impedance in congestive heart failure: current options and clinical applications. Am Heart J. 2009;157(3):402-411.
doi pubmed - Volodarskiy A, Nazeer H, Rosen L, Patel A, Jermyn R. Thoracic impedance & pulmonary artery pressure monitoring in prevention of heart failure hospitalizations. J Card Fail. 2017;23:S88.
doi - Perego GB, Oldani M, Pellegrini D, Brasca FMA, Malfatto G, Villani A, Brambilla R, et al. Correlation between pulmonary artery pressure and thoracic impedance: Insights from daily monitoring through an implanted device in chronic heart failure. Int J Cardiol. 2017;245:196-200.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


