| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 14, Number 2, April 2023, pages 106-114
Triggers of Atrial Fibrillation in the Geriatric Medical Intensive Care Unit: An Observational Study
Khaled Alya, c , Maram Shaatb, Sarah Hamzab, Safaa Alib
aCardiology Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt
bGeriatrics and Gerontology Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt
cCorresponding Author: Khaled Aly, Cardiology Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt
Manuscript submitted December 31, 2022, accepted February 13, 2023, published online March 25, 2023
Short title: Atrial Fibrillation and Trace Elements
doi: https://doi.org/10.14740/cr1461
| Abstract | ▴Top |
Background: Atrial fibrillation (AF) is a common arrhythmia in the non-cardiac intensive care unit (ICU). However, data concerning AF incidence and predictors in such populations are scarce and controversial. The study aimed to investigate the contributing factors of new-onset AF in elderly patients within the medical intensive care setting.
Methods: Patients admitted to ICU during a 6-month period were prospectively studied. Patients admitted for short period postoperative monitoring and patients with chronic or paroxysmal AF were excluded. The conditions involved as AF risk factors or “triggers” from demographic data, history, and echocardiography were recorded. Acute Physiology and Chronic Health Evaluation II score was calculated. Electrolytes including some trace elements (zinc, copper, and magnesium) were analyzed.
Results: The study included 142 patients (49% females). Mean age was 69.5 ± 7.3 years. AF was observed in 12%. Diagnosis of pneumonia (P < 0.001), low copper (P < 0.0001) and low zinc levels (P < 0.0001) was significantly associated with the occurrence of AF. By multivariate analysis, they remained statistically significant (odds ratio, 7.0; 95% confidence interval, 2.0 - 24.6; P < 0.01).
Conclusions: A significant fraction of ICU elderly patients manifests AF. The relevant factors contributing to AF incidence in the elderly are pneumonia and low zinc and low copper.
Keywords: Atrial fibrillation; Elderly; Trace elements; ICU
| Introduction | ▴Top |
Atrial fibrillation (AF) is a common cardiac arrhythmia in the intensive care unit (ICU) associated with increased morbidity and mortality [1]. AF occurs in 1.8-10% of non-cardiac patients admitted to the ICU [2]; in one study, it was approximately 5.3% [3] in surgical ICU patients, and in another 7.8% [4]. ICU mortality in septic shock patients with new-onset AF was 44% [4]. AF is more common in older patients [5]. With age, changes in the conduction system, atrial volume, and shape due to atrophy and fibrosis lead to a higher incidence of AF in older adults [6].
In critically ill patients, AF may develop due to fluid imbalances, electrolyte imbalances, hormonal imbalances, arrhythmogenic drugs, and systemic inflammatory responses [7].
Trace elements such as copper, cobalt, and arsenic may contribute to myocardial dysfunction; zinc deficiency may increase the susceptibility of the phospholipid cell membrane to free-radical damage and oxidative changes. Magnesium is an important determinant of the resting membrane potential of cardiac cell membranes, and potassium regulates heartbeat [8]. Imbalance in serum electrolyte levels such as potassium, sodium, calcium, and magnesium may result in arrhythmias [9].
Few studies tackled the subject of AF incidence and relationship with trace elements, but most of these studies worked either in cardiac surgery patients [10, 11] or non-cardiac surgery patients [5, 12], while data in general medical patients are limited. Those worked on general medical patients focused on sepsis patients [13, 14], severity of illness [15, 16], medications [3, 17], and interventions like catecholamine use and heart catheterization [18].
We intended to work with critically ill elderly patients to reveal risk factors for AF within the geriatric medical intensive care setting with vigilance to some trace elements.
| Materials and Methods | ▴Top |
This was a prospective, observational, single-center cross-sectional study to investigate the contributing factors of new-onset AF in elderly patients within a medical intensive care setting. All data were collected after detailed information was given to the patient and a written consent was obtained. The study was approved by the local Institutional Ethical Committee of Faculty of Medicine, Ain Shams University. This study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
The study recruited all elderly patients admitted to the ICU in a period of 6 months (May to October), and they were subdivided into two groups. The first group included patients with incident AF (patients who developed new-onset AF). The second group included patients who remained in sinus rhythm. Patients with the following conditions were excluded: patients admitted for brief monitoring (less than 24 h), patients with chronic AF, patients with known history of paroxysmal AF and those with implanted permanent or transient pacemaker.
Diagnosis of new onset AF
At baseline and during follow-up examination, 10-s 12-lead electrocardiography (ECG) was recorded with an ECG recorder (Bionet CardioCare).
All subjects underwent thorough history taking and physical examination at baseline.
Cardiovascular evaluation was performed as well as echocardiography to evaluate left atrial volume and dimensions, left ventricular ejection fraction, diastolic dysfunction, right and left ventricular systolic pressure.
ICU outcome measures
Acute Physiology and Chronic Health Evaluation II (APACHE II) score
It provides a classification of the severity of disease condition for adults admitted to ICU, and it is calculated from 12 routine physiologic measurements made during the first day after admission. An increasing score (range 0 to 71) correlates with the later risk of hospital death [16].
Systemic inflammatory response syndrome (SIRS)
It is an inflammatory disease that affects the entire body as the immune system responds to infection. SIRS is sensitive because approximately more than 90% of ICU patients meet SIRS criteria. Manifestations of SIRS include body temperature below 36 °C or above 38 °C, heart rate above 90 beats per minute, shortness of breath above 20 breaths per minute, white blood cell count below 4,000 cells/mm3 or above 12,000 cells/mm3. If two or more of these criteria are met, a patient is diagnosed with SIRS regardless of evidence of infection [19].
Laboratory test
It included complete blood count (CBC), serum chemistry (electrolytes/liver function test/kidney function test), and coagulation profile.
Statistical methods
Descriptive analysis was represented in means ± standard deviation (SD), minimum and maximum of the range for quantitative parametric data, and as numbers and percentages for the qualitative ones. In case of two independent groups with parametric quantitative data, inferential analyses were done using independent t-test. Inferential analyses for independent variables were done using Chi-square test for differences between proportions. The level of significance was taken at 95% (P < 0.05).
Multivariate analysis using a logistic regression model was done to evaluate significant contributing risk factors for new-onset AF.
| Results | ▴Top |
One hundred sixty-nine patients were admitted to the ICU during the 6 months period, and 27 were excluded due to early death or history of AF. Of the 142 remaining, 18 were converted to AF as shown in Figure 1.
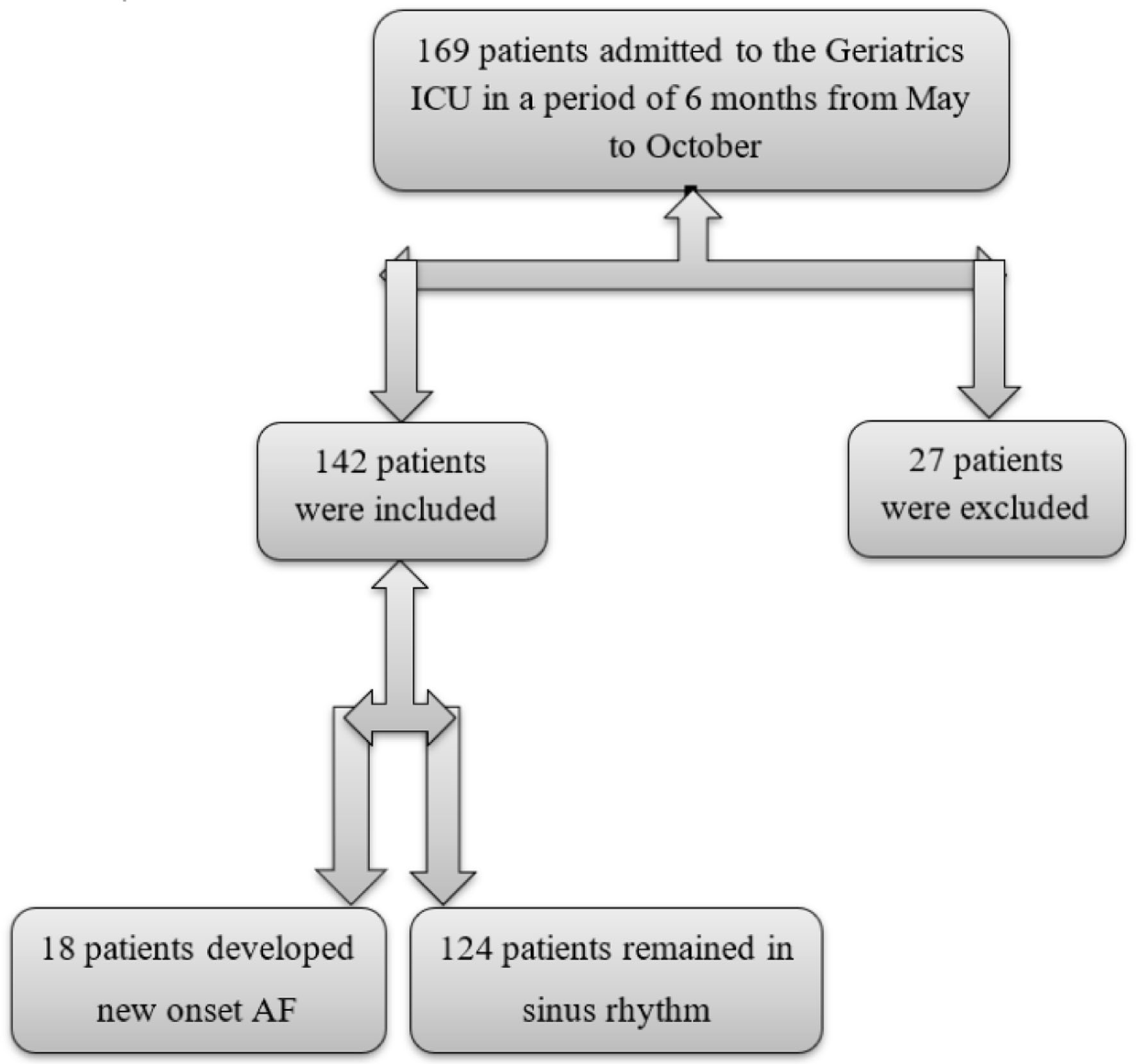 Click for large image | Figure 1. Study flow chart. |
Demographic data and baseline characteristics
The mean age of the sample was 69.5 ± 7.3 years (mean ± SD); males are nearly equal to females. Non-smokers represent two-thirds. The body mass index (BMI) of the sample was average. Comorbidities are shown in Table 1. There was no difference between sinus patients and incident AF patients regarding demographics, comorbidities and clinical presentation except for percentage of patients presented with pneumonia (50% in patients with incident AF vs. 9.6% in those who stayed in sinus rhythm, P ≤ 0.05).
 Click to view | Table 1. Demographic Data, Comorbidities and Clinical Presentation |
Clinical characteristics according to the onset of AF
Comparing different clinical characteristics, there was no statistically significant difference between both groups as regards ischemic heart disease (IHD), heart failure, hypertension, chronic obstructive airway disease (COPD), pulmonary embolism, obstructive sleep apnea, diabetes mellitus (DM), chronic liver disease, renal impairment, and hemorrhage. Similarly, there was no significant difference between those who developed AF and those who remained in sinus rhythm regarding APACHEII score, SIRS, or presence of septic shock, as shown in Table 2.
 Click to view | Table 2. Clinical Characteristics According to the Onset of Atrial Fibrillation |
Medications used
Studying different medications used among the study population with emphasis on beta-blockers (BBs, 16% vs. 15%, P = 1) (bisoprolol was the main prescribed one), calcium channel blockers (CCBs, 11% vs. 8%, P = 0.65) as well as inotropic and vasopressor agents (11% vs. 12%, P = 1), there was no significant difference between those who developed AF and those who remained in sinus rhythm (Fig. 2).
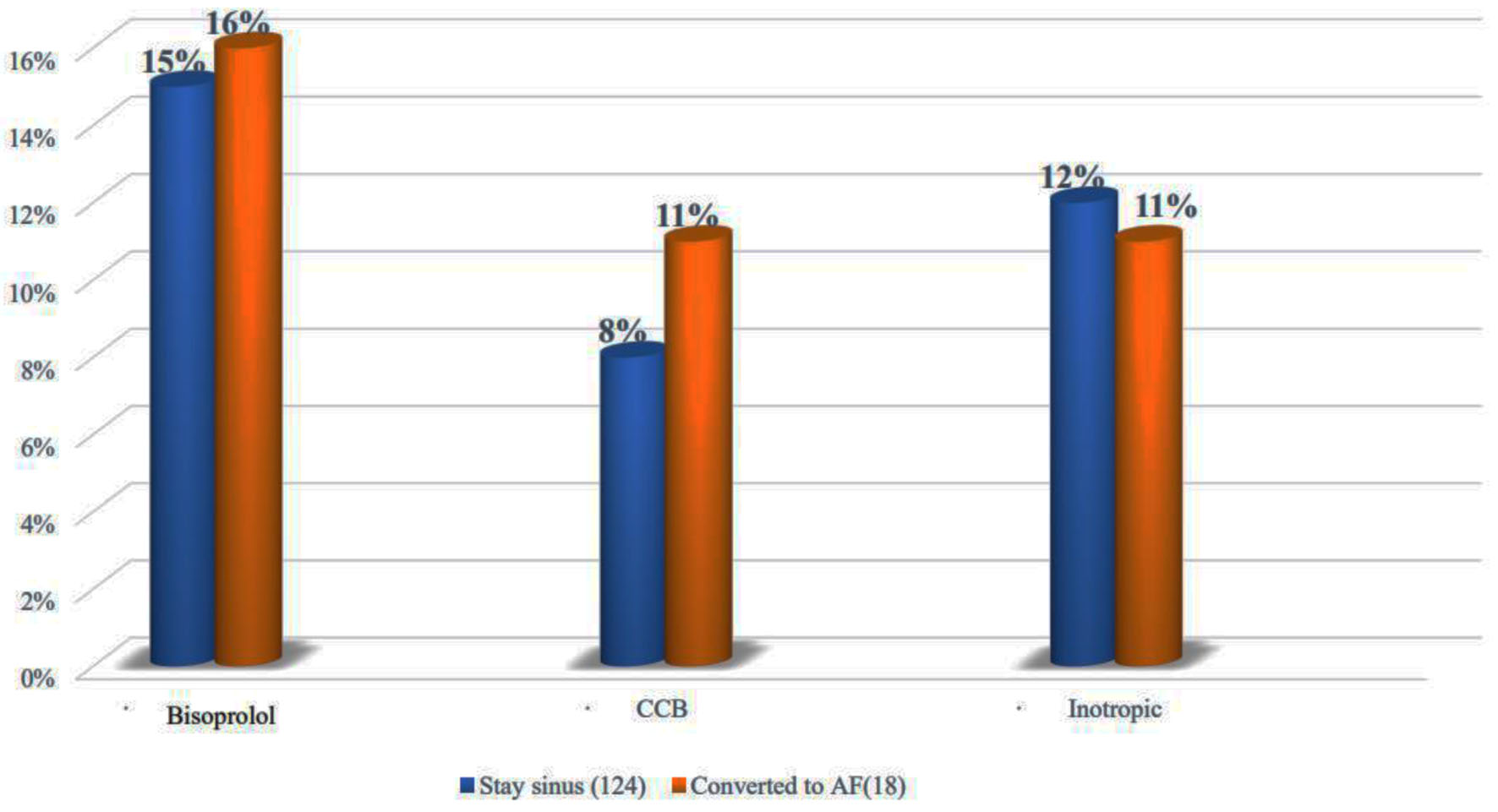 Click for large image | Figure 2. Comparison between relevant medications used in the sinus and the AF groups. AF: atrial fibrillation. |
Baseline serum electrolyte concentrations
Comparing various serum electrolyte concentrations at baseline between those who remained sinus and those who developed new-onset AF, we found no significant difference between all except copper and zinc levels. There was a statistically significant lower serum level of copper (128 ± 64 µg/dL vs. 72.3 ± 37.4 µg/dL, P < 0.0001) and zinc (132 ± 79 µg/dL vs. 52 ± 27.5 µg/dL, P < 0.0001) in patients converted to AF (Table 3).
 Click to view | Table 3. Baseline Serum Electrolyte Concentrations |
Laboratory results
There was no significant difference between both groups regarding other laboratory results as shown in Table 4.
 Click to view | Table 4. Comparison of Laboratory Results |
Multivariate logistic regression model for factors increasing the incidence of AF in critically ill elderly patients
We performed logistic regression analysis of occurrence of pneumonia, serum copper at baseline and serum zinc at baseline as predictors of developing new-onset AF in critically ill patients admitted to geriatric ICU (Table 5).
 Click to view | Table 5. Logistic Regression for Factors Increasing the Incidence of AF in Critically Ill Elderly Patients |
Pneumonia had a coefficient of 0.254 that was statistically significant with P value < 0.001.
As well as copper and zinc, they had a statistically highly significant coefficient ((-0.252 and -0.268, respectively) with P < 0.0001.
| Discussion | ▴Top |
The aim of the study was to measure the incidence rate of AF in elderly patients admitted to non-cardiac medical intensive care units and its possible risk factors. The study revealed that the incidence rate of AF in elderly patients was 12.6%. In the current study, we found that the incidence of AF is associated with pneumonia diagnosis, low serum copper and low serum zinc in univariate and multivariate analysis. Other electrolytes and possible triggers (like APACHE II score, inotropic agents use, SIRS and abnormal vital signs) were not a significant precipitant to AF in the present study.
In our study, APACHE II, SIRS and septic shock were not risk factors for AF. Yet there was a trend of higher percentage of patients having septic shock in the AF group (27% compared to 8% in sinus group). In the studies by Della et al and Sanne et al, SIRS and septic shock were risk factors of AF, and this is mostly due to atypical presentation of infection in elderly which includes absence of fever, leukocytosis as well as limited number of patients in the current study [20, 21].
As regards new-onset AF, the incidence varied widely according to the setting. It is reported to be a high rate among septic shock patients (46.0%) [22]. Postoperative AF has been reported to occur in 15-50% of patients with cardiac surgery [23, 24] and 10-42% with thoracic surgery [25, 26], suggesting that these two specific cohorts might have a higher incidence of AF compared with the general ICU population. The incidence range in our study (12.6%) is higher than previously thought (4-9%) [27], possibly due to the aging population and the development of more complex medical comorbidities.
Trace elements such as copper and zinc have been studied in relation to AF. Two studies have been performed on postoperative AF [28, 29]. First, the concentrations of these elements in the blood plasma of patients undergoing elective coronary artery bypass grafting were determined. Blood samples were collected during and after cardiopulmonary bypass. Plasma concentrations of copper, zinc, and magnesium were measured. AF occurred in 21.7% of patients, and there were significant differences in zinc and copper concentrations between postoperative day 1 and day 3 between those who developed AF and who did not. Zinc concentrations recovered more slowly in patients with AF after cardiopulmonary bypass surgery than in patients without AF [28]. Another study also applied to patients undergoing cardiopulmonary bypass surgery. It has been shown that patients presenting with heart failure early after perfusion have significantly lower blood copper and zinc concentrations than patients without complications postoperatively [30]. In contrast, Negreva et al in 2015 studied so far 33 patients with paroxysmal AF and 33 controls without evidence of arrhythmia. The plasma levels of the elements were determined three times in patients: immediately after hospitalization (initial value), 24 h and 28 days after restoration of sinus rhythm, and in the control group. In patients with paroxysmal AF, plasma zinc concentrations did not differ from controls, and no signs of arrhythmias were reported. They concluded that the lack of dynamics in the index values gives us good reason to believe that zinc is not associated with disease onset and recurrence [30]. Zinc has antioxidant and anti-inflammatory properties [31, 32]. In recent years, research into the mechanisms of AF has revealed the development of oxidative stress and inflammation in patients with this disease [33, 34].
Regarding copper, the changes in its homeostasis are considered an independent risk factor for cardiovascular death [34, 35]. Negreva et al in 2014 assessed copper status early in paroxysmal AF and monitored changes after restoration of sinus rhythm. They found that copper levels decreased during the first few hours of the arrhythmia and subsequently increased after restoration of sinus rhythm, suggesting that these changes are closely related to the pathogenesis of paroxysmal AF [34].
Regarding magnesium, it is an important cofactor for the Na-K-ATP pump, which controls the flow of sodium and potassium across cell membranes [35]. Disturbance or change of the function of this pump during hypomagnesemia impairs myocardial excitability. Magnesium also prolongs the effective refractory period and disrupts the function of inwardly rectifying potassium channels, although not all studies have shown this effect [36-39]. Magnesium infusions prolong atrioventricular nodal conduction time [40], while low serum magnesium concentrations increase sinoatrial node automaticity [41]. Clinical studies have shown that intravenous magnesium can improve the control of AF and help maintain sinus rhythm [42]. Conversely, hypomagnesemia increases the digoxin dose required for control [43] and lowers the threshold for digoxin-related arrhythmias [44].
Some studies have described the relationship between hypomagnesemia and AF risk in the postoperative state after cardiac surgeries. Another study demonstrated this association in a broader community-based cohort [44].
In the current study, baseline serum potassium was normal and hence magnesium so both were unrelated to incident AF in our data.
In the current study, prior use of some medications such as BBs (bisoprolol) and CCBs as well as the use of inotropes and vasopressors was not associated with increased risk of AF, and this is mostly because of the usage of norepinephrine more than dopamine which is less arrhythmogenic.
Although the study of Gennaro et al showed that BBs significantly reduce the incidence of post-operative AF [45], also in the study of Imad et al, BBs appear to effectively prevent occurrence of AF in patients with systolic heart failure, which is a longitudinal study [46]. Also the study of Sacha et al showed that dopamine use may be associated with a higher incidence of arrhythmias compared with norepinephrine administration [47].
Indeed, the early detection of trace elements allows not only making a general conclusion that they have relation to the pathogenesis of AF, but also in particular that it may be a part of the initiation mechanisms. In the current study while copper and zinc were lower in the AF group an association was not established and thus further studies are needed before one proceed to replace copper and zinc on a routine basis to prevent AF.
Study limitations
Being a single-center study without longer term follow-up and relatively limited number of participants are the main limitations of this study. A larger number of enrolled patients can result in higher number of AF patients to be studied in future studies.
Conclusions
We can conclude that pneumonia and baseline deficiency of copper and zinc are independent predictors of development of new-onset AF in critically ill patients admitted to geriatric ICU. This finding will help to routinely check zinc and copper serum levels at baseline and try to avoid such electrolyte imbalance.
Acknowledgments
We acknowledge the geriatric ICU for kindly providing technical support and ABG set for blood gas analyses.
Financial Disclosure
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
No data were collected before detailed information was given to the patient and a written consent was obtained.
Author Contributions
KA conceptualized the project, reviewed the literature, collected the data, analyzed the data, and wrote the manuscript. SA conceptualized the project and collected the data. MS conceptualized the project, reviewed the literature, collected the data, and analyzed the data. SH conceptualized the project and reviewed the literature. All authors read and approved the final manuscript.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Abbreviations
AF: atrial fibrillation; ALT: alanine transaminase; APACHE II: Acute Physiology and Chronic Health Evaluation II; BBs: beta-blockers; BMI: body mass index; BUN: blood urea nitrogen; CBC: complete blood count; CCBs: calcium channel blockers; COPD: chronic obstructive pulmonary disease; ECG: electrocardiography; ICU: intensive care unit; K: potassium; Na: sodium; SIRS: systemic inflammatory response syndrome; WBC: white blood cell count
| References | ▴Top |
- Arrigo M, Bettex D, Rudiger A. Management of atrial fibrillation in critically ill patients. Crit Care Res Pract. 2014;2014:840615.
doi pubmed pmc - Kanji S, Williamson DR, Yaghchi BM, et al. Epidemiology and management of atrial fibrillation in medical and non-cardiac surgical adult intensive care unit patients. Journal of Critical Care. 2012;27(3):326-401.
doi pubmed - Seguin P, Signouret T, Laviolle B, Branger B, Malledant Y. Incidence and risk factors of atrial fibrillation in a surgical intensive care unit. Crit Care Med. 2004;32(3):722-726.
doi pubmed - Meierhenrich R, Steinhilber E, Eggermann C, Weiss M, Voglic S, Bogelein D, Gauss A, et al. Incidence and prognostic impact of new-onset atrial fibrillation in patients with septic shock: a prospective observational study. Crit Care. 2010;14(3):R108.
doi pubmed pmc - Brathwaite D, Weissman C. The new onset of atrial arrhythmias following major noncardiothoracic surgery is associated with increased mortality. Chest. 1998;114(2):462-468.
doi pubmed - Almassi GH, Schowalter T, Nicolosi AC, Aggarwal A, Moritz TE, Henderson WG, Tarazi R, et al. Atrial fibrillation after cardiac surgery: a major morbid event? Ann Surg. 1997;226(4):501-511; discussion 511-503.
doi pubmed pmc - Artucio H, Pereira M. Cardiac arrhythmias in critically ill patients: epidemiologic study. Crit Care Med. 1990;18(12):1383-1388.
doi pubmed - Yan YQ, Liu XC, Jing WB, Bai XY, Yang Q, He GW. Alterations of Zinc, Copper, and Magnesium Concentrations During and After Open Heart Surgery. The FASEB Journal. 2012;26(1 Supplement):1103-1126.
doi - Schroll A. Importance of magnesium for the electrolyte homeostasis. Advances in magnesium research: magnesium in cardiology: proceedings of the 5th European Congress on Magnesium. 1997;463-472.
- Mathew JP, Fontes ML, Tudor IC, Ramsay J, Duke P, Mazer CD, Barash PG, et al. A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA. 2004;291(14):1720-1729.
doi pubmed - Al-Khatib SM, Hafley G, Harrington RA, Mack MJ, Ferguson TB, Peterson ED, Califf RM, et al. Patterns of management of atrial fibrillation complicating coronary artery bypass grafting: Results from the PRoject of Ex-vivo Vein graft ENgineering via Transfection IV (PREVENT-IV) Trial. Am Heart J. 2009;158(5):792-798.
doi pubmed - von Knorring J, Lepantalo M, Lindgren L, Lindfors O. Cardiac arrhythmias and myocardial ischemia after thoracotomy for lung cancer. Ann Thorac Surg. 1992;53(4):642-647.
doi pubmed - Christian SA, Schorr C, Ferchau L, Jarbrink ME, Parrillo JE, Gerber DR. Clinical characteristics and outcomes of septic patients with new-onset atrial fibrillation. J Crit Care. 2008;23(4):532-536.
doi pubmed - Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270(24):2957-2963.
doi pubmed - Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13(10):818-829.
doi pubmed - Arora S, Lang I, Nayyar V, Stachowski E, Ross DL. Atrial fibrillation in a tertiary care multidisciplinary intensive care unit—incidence and risk factors. Anaesth Intensive Care. 2007;35(5):707-713.
doi pubmed - Walkey AJ, Wiener RS, Ghobrial JM, Curtis LH, Benjamin EJ. Incident stroke and mortality associated with new-onset atrial fibrillation in patients hospitalized with severe sepsis. JAMA. 2011;306(20):2248-2254.
doi pubmed pmc - American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20(6):864-874.
doi - Echahidi N, Pibarot P, O'Hara G, Mathieu P. Mechanisms, prevention, and treatment of atrial fibrillation after cardiac surgery. J Am Coll Cardiol. 2008;51(8):793-801.
doi pubmed - Ayed SD, Ayed S, Atig R, Tilouche N, Ali HBS, Gharbi R, Hassen MF, et al. Service de Reanimation Medicale, EPS Taher Sfar Mahdia, Mahdia, Tunisie. 2012.
- Kuipers S, Klein Klouwenberg PM, Cremer OL. Incidence, risk factors and outcomes of new-onset atrial fibrillation in patients with sepsis: a systematic review. Crit Care. 2014;18(6):688.
doi pubmed pmc - January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, Jr., Conti JB, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):2071-2104.
doi pubmed - Rivero-Ayerza M, Scholte Op Reimer W, Lenzen M, Theuns DA, Jordaens L, Komajda M, Follath F, et al. New-onset atrial fibrillation is an independent predictor of in-hospital mortality in hospitalized heart failure patients: results of the EuroHeart Failure Survey. Eur Heart J. 2008;29(13):1618-1624.
doi pubmed - Seguin P, Launey Y. Atrial fibrillation is not just an artefact in the ICU. Crit Care. 2010;14(4):182.
doi pubmed pmc - Yan YQ, Zou LJ. Relation between zinc, copper, and magnesium concentrations following cardiopulmonary bypass and postoperative atrial fibrillation in patients undergoing coronary artery bypass grafting. Biol Trace Elem Res. 2012;148(2):148-153.
doi pubmed - Nuutinen LS, Ryhanen P, Pihlajaniemi R, Hollmen A, Tyrvainen L. The levels of zinc, copper, calcium and magnesium in serum and urine after heart-valve replacement. Effects of oxygenator type and postoperative parenteral nutrition. Infusionsther Klin Ernahr. 1981;8(5):214-217.
doi pubmed - Negreva MN, Penev AP, Georgiev S, Aleksandrova AA. Paroxysmal atrial fibrillation: dynamics of the main antioxidant enzymes—superoxide dismutase and catalase. Folia Med (Plovdiv). 2014;56(2):96-101.
doi pubmed - Prasad AS. Zinc: an antioxidant and anti-inflammatory agent: role of zinc in degenerative disorders of aging. J Trace Elem Med Biol. 2014;28(4):364-371.
doi pubmed - Zago MP, Oteiza PI. The antioxidant properties of zinc: interactions with iron and antioxidants. Free Radic Biol Med. 2001;31(2):266-274.
doi pubmed - Friedrichs K, Klinke A, Baldus S. Inflammatory pathways underlying atrial fibrillation. Trends Mol Med. 2011;17(10):556-563.
doi pubmed - Yang KC, Dudley SC, Jr. Oxidative stress and atrial fibrillation: finding a missing piece to the puzzle. Circulation. 2013;128(16):1724-1726.
doi pubmed pmc - Engstrom G, Hedblad B, Tyden P, Lindgarde F. Inflammation-sensitive plasma proteins are associated with increased incidence of heart failure: a population-based cohort study. Atherosclerosis. 2009;202(2):617-622.
doi pubmed - Leone N, Courbon D, Ducimetiere P, Zureik M. Zinc, copper, and magnesium and risks for all-cause, cancer, and cardiovascular mortality. Epidemiology. 2006;17(3):308-314.
doi pubmed - Negreva MN, et al. Assessment of copper status in patients with paroxysmal atrial fibrillation. J Cardiobiol. 2014;2(2):5.
doi - Skou JC, Butler KW, Hansen O. The effect of magnesium, ATP, P i , and sodium on the inhibition of the (Na + + K + )-activated enzyme system by g-strophanthin. Biochim Biophys Acta. 1971;241(2):443-461.
doi pubmed - Stark G, Stark U, Pilger E, Honigl K, Bertuch H, Tritthart HA. The influence of elevated Mg2+ concentrations on cardiac electrophysiologic parameters. Cardiovasc Drugs Ther. 1989;3(2):183-189.
doi pubmed - DiCarlo LA, Jr., Morady F, de Buitleir M, Krol RB, Schurig L, Annesley TM. Effects of magnesium sulfate on cardiac conduction and refractoriness in humans. J Am Coll Cardiol. 1986;7(6):1356-1362.
doi pubmed - Op't Hof T, Mackaay AJ, Bleeker WK, Jongsma HJ, Bouman LN. Differences between rabbit sinoatrial pacemakers in their response to Mg, Ca and temperature. Cardiovasc Res. 1983;17(9):526-532.
doi pubmed - Onalan O, Crystal E, Daoulah A, Lau C, Crystal A, Lashevsky I. Meta-analysis of magnesium therapy for the acute management of rapid atrial fibrillation. Am J Cardiol. 2007;99(12):1726-1732.
doi pubmed - DeCarli C, Sprouse G, LaRosa JC. Serum magnesium levels in symptomatic atrial fibrillation and their relation to rhythm control by intravenous digoxin. Am J Cardiol. 1986;57(11):956-959.
doi pubmed - Foroulis CN, Kotoulas C, Lachanas H, Lazopoulos G, Konstantinou M, Lioulias AG. Factors associated with cardiac rhythm disturbances in the early post-pneumonectomy period: a study on 259 pneumonectomies. Eur J Cardiothorac Surg. 2003;23(3):384-389.
doi pubmed - Seller RH, Cangiano J, Kim KE, Mendelssohn S, Brest AN, Swartz C. Digitalis toxicity and hypomagnesemia. Am Heart J. 1970;79(1):57-68.
doi pubmed - Zaman AG, Alamgir F, Richens T, Williams R, Rothman MT, Mills PG. The role of signal averaged P wave duration and serum magnesium as a combined predictor of atrial fibrillation after elective coronary artery bypass surgery. Heart. 1997;77(6):527-531.
doi pubmed pmc - Khan AM, Lubitz SA, Sullivan LM, Sun JX, Levy D, Vasan RS, Magnani JW, et al. Low serum magnesium and the development of atrial fibrillation in the community: the Framingham Heart Study. Circulation. 2013;127(1):33-38.
doi pubmed pmc - Amato B, Compagna R, Coretti G, Vigliotti G, Rocca A, Fappiano F, Rossi R, et al. Abstracts from the 26th National Congress of the Italian Society of Geriatric Surgery, 19-22 June 2013, Naples, Italy. BMC Surg. 2013;13(Suppl 1):A1-A20.
doi pubmed pmc - Nasr IA, Bouzamondo A, Hulot JS, Dubourg O, Le Heuzey JY, Lechat P. Prevention of atrial fibrillation onset by beta-blocker treatment in heart failure: a meta-analysis. Eur Heart J. 2007;28(4):457-462.
doi pubmed - Pollard S, Edwin SB, Alaniz C. Vasopressor and inotropic management of patients with septic shock. P T. 2015;40(7):438-450.
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


