| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Original Article
Volume 9, Number 4, August 2018, pages 224-230
Ivabradine Versus Beta-Blockers in Mitral Stenosis in Sinus Rhythm: An Updated Meta-Analysis of Randomized Controlled Trials
John Daniel A. Ramosa, Elleen L. Cunanana, Lauro L. Abrahan, IVa, Marc Denver A. Tiongsona, b, Felix Eduardo R. Punzalana
aDepartment of Medicine, Section of Cardiology, University of the Philippines, Philippine General Hospital, Manila, Philippines
bCorresponding Author: Marc Denver A. Tiongson, Section of Cardiology, University of the Philippines, Philippine General Hospital, Manila, Philippines
Manuscript submitted May 31, 2018, accepted June 12, 2018
Short title: Ivabradine vs. Beta-Blockers in MS
doi: https://doi.org/10.14740/cr737w
| Abstract | ▴Top |
Background: Symptoms of mitral stenosis (MS) are worsened during tachycardia and exercise. Beta-blockers are used in controlling heart rate (HR) in MS, resulting in symptom improvement, but coming with significant side effects. Ivabradine has a selective action on the sinus node devoid of the usual side effects of beta-blockers. Small studies have recently investigated the role of ivabradine in MS in sinus rhythm. Our aim was to determine the efficacy of ivabradine, compared to beta-blockers, in terms of exercise duration, maximum HR achieved, resting HR, mean gradient, and working capacity among patients with MS in sinus rhythm.
Methods: We conducted a systematic search of studies using MEDLINE, Google Scholar, ScienceDirect, Scopus, Clinical Key, Cochrane, and clinicaltrials.gov databases in all languages and examined reference lists of studies. We included studies if they are: 1) randomized controlled trials comparing ivabradine and beta-blockers; 2) of adults ≥ 19 years old with MS in sinus rhythm; and 3) reported data on exercise duration, maximum HR achieved, resting HR, mean gradient, and working capacity. Studies identified were assessed for risk of bias using the Cochrane Collaboration Tool for Assessing Risk of Bias. We used inverse variance analysis of fixed effects to compute for mean difference, carried out using Review Manager (RevMan) 5.3.
Results: Pooled analysis from five identified trials showed that among patients with MS in sinus rhythm, ivabradine was better compared to beta-blockers in total exercise duration (mean difference: 32.73 s (95% CI: 12.19, 53.27; P = 0.002; I2 = 0%)), maximum HR achieved after exercise (mean difference: -3.87 beats per minute (95% CI: -5.88, -1.860; P = 0.0002; I2 = 23%)), and work capacity (mean difference: 0.56 METS (95% CI: 0.33, 0.80; P < 0.00001; I2 = 0%)); inferior to beta-blockers in resting HR achieved (mean difference: 1.83 s (95% CI: 0.39, 3.28; P = 0.01; I2 = 91%)); and comparable to beta-blockers in terms of mean gradient (mean difference: -0.52 mm Hg (95% CI: -1.20, 0.16; P = 0.13; I2 = 6%)).
Conclusions: Ivabradine is better or comparable to beta-blockers in terms of the outcomes measured, and may be considered as an alternative for patients with MS in sinus rhythm who are intolerant to beta-blockers.
Keywords: Ivabradine; Mitral stenosis
| Introduction | ▴Top |
Rheumatic heart disease (RHD) remains a significant health burden worldwide [1]. Carapetis estimated that there are 15 million cases worldwide, with 282,000 new cases and 233,000 annual deaths [2]. About 15% of all patients with heart failure in endemic countries are due to RHD [3]. As late as 2008, the prevalence of RHD continued to increase in almost all of the World Health Organization (WHO) regions, with the highest burden to be found in Southeast Asia, numbering 50 in 1000 persons affected [1]. In the Philippines, Jara and Tumabiene cited a prevalence of 0.8 - 1.3/1000 in a pediatric series of 10-year duration [4].
Mitral stenosis (MS), with mitral valve area ≤ 2 cm2, is the most common valve affectation of RHD. Approximately 25% of all patients with RHD have isolated MS, and approximately 40% have combined MS and MR [5]. MS results to an obstructed flow from the left atrium to the left ventricle, with consequent increase in transvalvular pressure gradient. The transvalvular pressure gradient for any given valve area is a function of the square of the transvalvular flow rate [5]. Thus a doubling of flow rate quadruples the pressure gradient. The elevated left atrial pressure, in turn, raises pulmonary venous and capillary pressures, resulting in exertional dyspnea [5]. Bouts of dyspnea in MS patients are precipitated by tachycardia. At any given stroke volume, tachycardia results in a higher instantaneous volume flow rate and higher transmitral pressure gradient, which elevate left atrial pressures further [5]. Heart rate (HR) control is thus of value and is reasonably recommended as therapy for patients with MS. In the 2014 ACC/AHA guidelines, HR control is a Class IIa-C recommendation for patients with MS and AF in rapid ventricular response and Class IIb-B for MS patients in sinus rhythm [6].
Beta-blockers have been shown in a number of trials to be effective as medical therapy in MS, though there are also conflicting data [7-10]. Another issue that besets beta-blockers in MS is the potential for adverse reactions, including fatigue, bronchoconstriction, AV blocks, depression, and intolerance [10]. In the ACC/AHA guidelines, beta-blockers, along with calcium antagonists, are choices for rate control in MS [6].
Ivabradine is a selective HR controller that acts selectively on the sinus node via the If, or funny current [11]. It has been found to have no effect on myocardial contractility, has very few side effects, and has better safety profile than beta-blockers [11]. It is recommended as a negative chronotropic agent for both stable angina [12] and heart failure [13], as supported by the SHIFT [14] and BEAUTIFUL [15] studies.
In recent years, small trials have evaluated the efficacy of ivabradine as an alternative rate controller to beta-blockers for MS in sinus rhythm. Agrawal has shown that ivabradine controls exertional symptoms more than metoprolol [16]. Similarly, Saggu showed that ivabradine was comparable to metoprolol in reducing symptoms and improving hemodynamics in these patients [17]. Parakh showed that ivabradine is more effective than atenolol in effort-related symptoms [18], but this was not the same for the study by Rajesh [19].
Adviento et al [20] performed a meta-analysis to assess the effect of ivabradine on symptoms and hemodynamic parameters in MS patients who are in sinus rhythm. A total of three small studies (one published, two unpublished) were included, comprising 121 patients, and showed that ivabradine was similar to beta-blockers in exercise duration time, HR achieved with exercise, and mean gradient.
In light of larger randomized trials that came out in the past 2 years, we decided to include new data and perform pooled analysis to further evaluate the role of ivabradine in this patient group.
Research question
Among patients with MS in sinus rhythm, is ivabradine comparable to beta-blockers in terms of exercise duration, maximum HR achieved, resting HR, mean gradient and working capacity?
Objective
Determine the efficacy of ivabradine, compared to beta-blockers, in terms of exercise duration, maximum HR achieved, resting HR, mean gradient, and working capacity among patients with MS in sinus rhythm.
| Methods | ▴Top |
Literature search
We conducted a systematic search of studies using MEDLINE, EMBASE, ScienceDirect, Scopus, Google Scholar, ClinicalKey, Cochrane Database of Systematic Reviews, clinicaltrials.gov, and Cochrane Central Register of Controlled Trials databases, with no language restrictions. We used the following search terms (in both free text and MESH strategies when using MEDLINE): “ivabradine”, “mitral stenosis”, “sinus rhythm”, “randomized controlled trial”, “mean gradient”, “heart rate”, and “exercise tolerance” or “work capacity”. A review of the reference lists of original and review article was also performed, as well as related links of the relevant publications. Studies were individually screened for eligibility and the full texts of relevant articles were obtained, when available. Authors of studies were contacted when there was no available full text.
Study selection
We included studies if they are: 1) randomized controlled trials; 2) with population that included adults ≥ 19 years old with MS in sinus rhythm; 3) compared ivabradine with beta-blockers; and 4) reported data on the following outcomes: exercise duration, mean gradient, exercise and/or resting HR, and work capacity. Four reviewing authors independently evaluated the eligibility of each study included in this meta-analysis. The validity and quality of each study was assessed using the Cochrane Tool for Assessing Risk of Bias for randomized trials. Disagreements were resolved by discussion and consensus.
Data collection and analysis
Information on patient and study characteristics, such as beta-blocker used and the pre-specified outcomes, were then extracted independently by two authors (JDR and ELC) using a standardized data collection table. We used inverse variance analysis of fixed effects to compute for mean difference between ivabradine and beta-blockers on the different outcomes, with 95% confidence intervals, and to generate forest plots. I2 test was used to assess heterogeneity. Funnel plot analysis was done to examine for publication bias. Analyses were carried out using Review Manager (RevMan) 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen).
| Results | ▴Top |
Search for studies and strategy
Our MEDLINE search yielded a total of seven potential articles. Search from other databases and from reference lists, and removing duplicate hits, yielded one additional study, which was an abstract of an unpublished study that was presented in a conference in India. There was no full text, and we were not able to obtain a full paper from the respective author. We evaluated a total of only eight titles and abstracts. Of these, three were ineligible for inclusion (one was a commentary, one was a letter to an editor, and one was a conference article on an abstract that had incomplete data). The full articles of the remaining articles were obtained and reviewed. All met the specified inclusion criteria. A summary of the search strategy is shown in Figure 1.
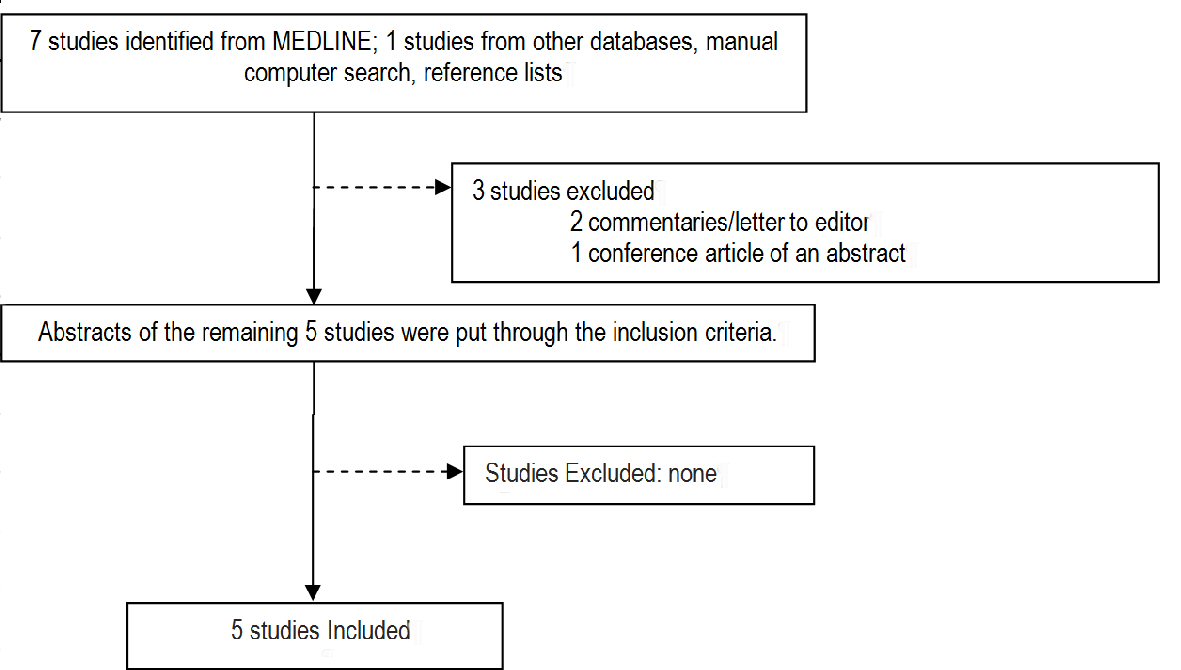 Click for large image | Figure 1. Summary of search strategy. |
Study characteristics and quality
Five studies, comprising a total of 362 patients, were included in this meta-analysis. Table 1 [16-19, 21] gives the summary of the study characteristics. All of the studies are small, with the largest study having 97 patients. Most involved patients with mild-to-moderate MS, with the exception of one study that had severe MS patients included. All are randomized trials, majority of which are open-label in design, and one with a cross-over design. The beta-blocker used was either metoprolol or atenolol. The studies ran for 4 weeks to 3 months. Figure 2 summarizes the risk of bias analysis. The studies of Agrawal [16] and Dhanger [21] have unclear risk of bias, while the rest have unclear-to-low risk of bias. Parakh’s [18] was graded high risk in participant blinding. Funnel plot analysis of the included studies did not reveal publication bias (Supplementary Fig. 1, www.cardiologyres.org).
 Click to view | Table 1. Characteristics of Included Studies |
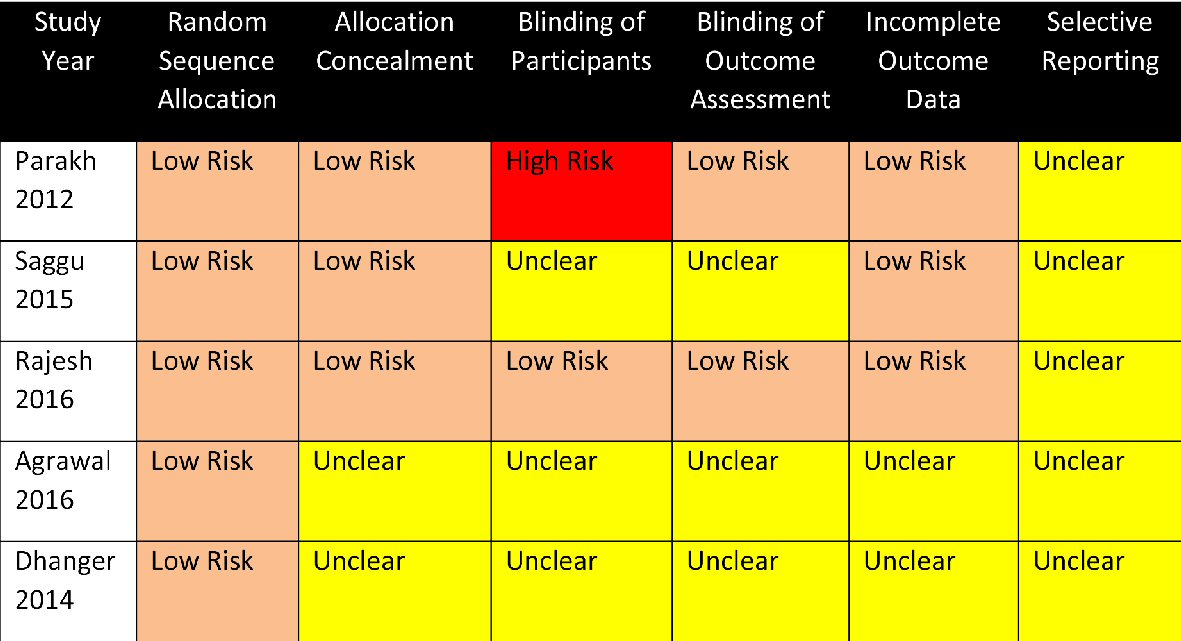 Click for large image | Figure 2. Summary of risk of bias assessment of the included studies. |
Ivabradine and mean time of exercise duration
Four studies looked at the effect of ivabradine to mean time of exercise duration (TED) and was found to be better than beta-blocker (mean difference: 32.73 s (95% CI: 12.19, 53.27; P = 0.002)) (Fig. 3). Data was homogenous (I2 = 0%).
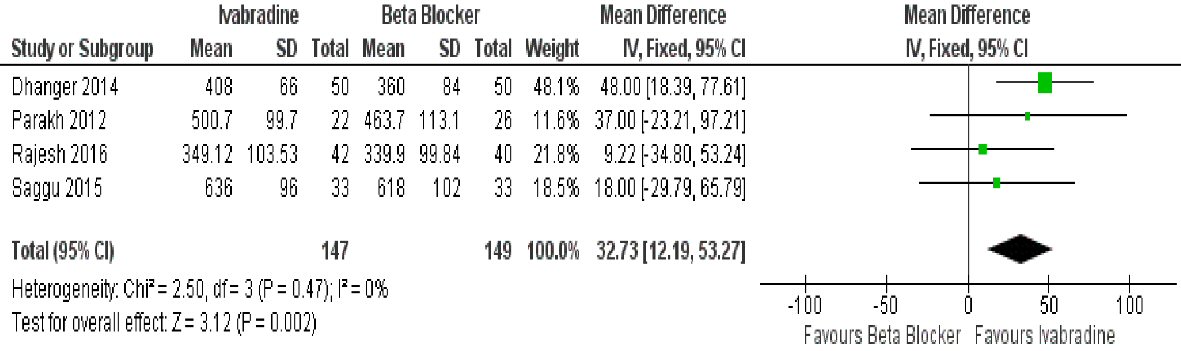 Click for large image | Figure 3. Forest plot showing the mean time of exercise duration with ivabradine versus beta-blocker in patients with MS in sinus rhythm. |
Ivabradine and maximal HR achieved after exercise
Three studies showed that ivabradine was better compared to beta-blocker in terms of maximal HR achieved after exercise (mean difference: -3.87 beats per minute (95% CI: -5.88, -1.860; P = 0.0002)) (Fig. 4). Heterogeneity was low at I2 = 23%.
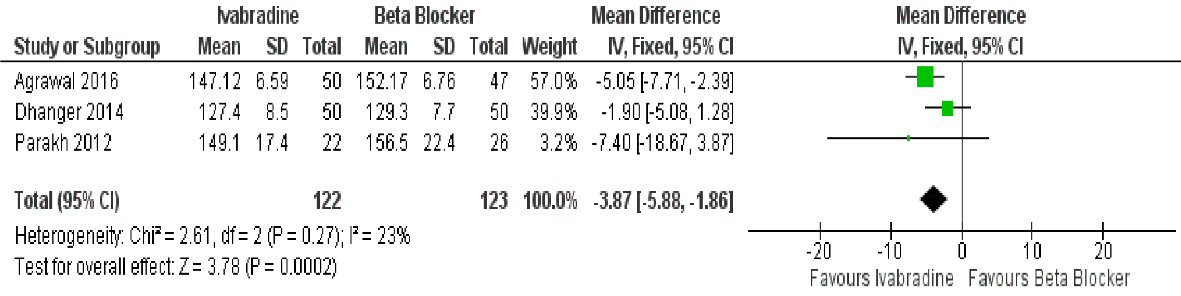 Click for large image | Figure 4. Forest plot showing the maximal HR achieved with ivabradine versus beta-blocker in patients with MS in sinus rhythm. |
Ivabradine and work capacity
Ivabradine was also found to be better compared to beta-blocker when it comes to improvement in work capacity (mean difference: 0.56 METS (95% CI: 0.33, 0.80; P < 0.00001)) (Fig. 5). Data was homogenous (I2 = 0%).
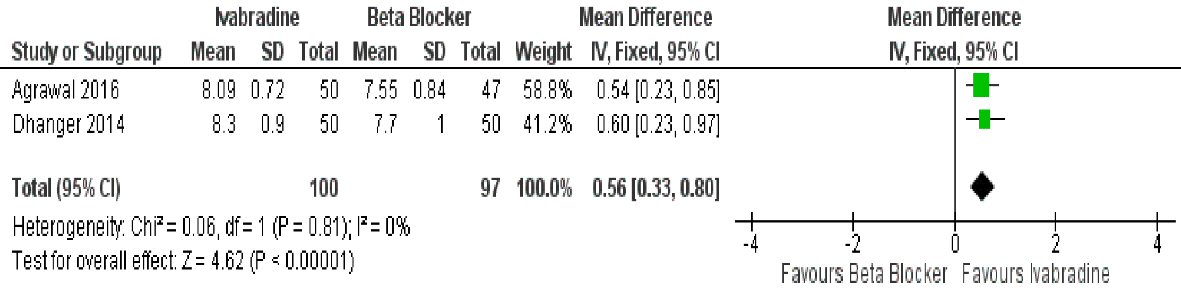 Click for large image | Figure 5. Forest plot showing the work capacity achieved with ivabradine versus beta-blocker in patients with MS in sinus rhythm. |
Ivabradine and mean gradient
Two studies had data on reduction of mean gradient with treatment, and showed that ivabradine was comparable to beta-blocker in this aspect (mean difference: -0.52 mm Hg (95% CI: -1.20, 0.16; P = 0.13)) (Fig. 6). Again, data had low heterogeneity (I2 = 6%).
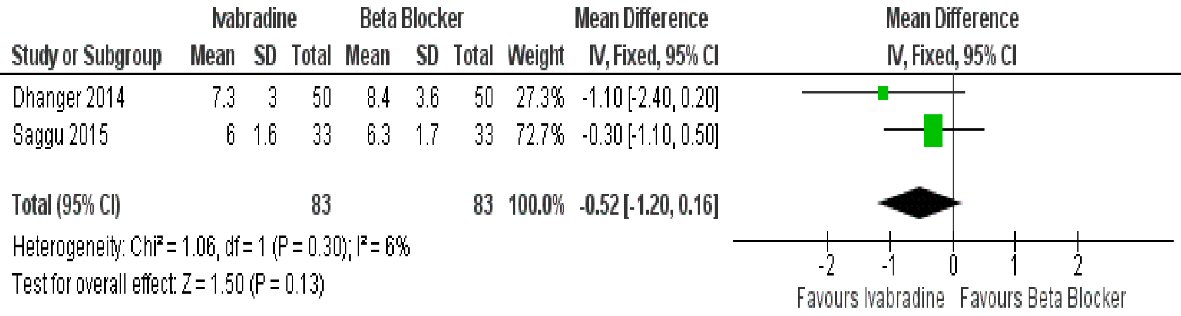 Click for large image | Figure 6. Forest plot showing the mean transvalvular gradient achieved with ivabradine versus beta-blocker in patients with MS in sinus rhythm. |
Ivabradine and resting HR
Three studies examined the effect of ivabradine in resting HR compared to beta-blocker, and showed that ivabradine was inferior to the latter in this regard (mean difference: 1.83 s, (95% CI: 0.39, 3.28; P = 0.01)) (Fig. 7). Data, however, was highly heterogenous (I2 = 91%).
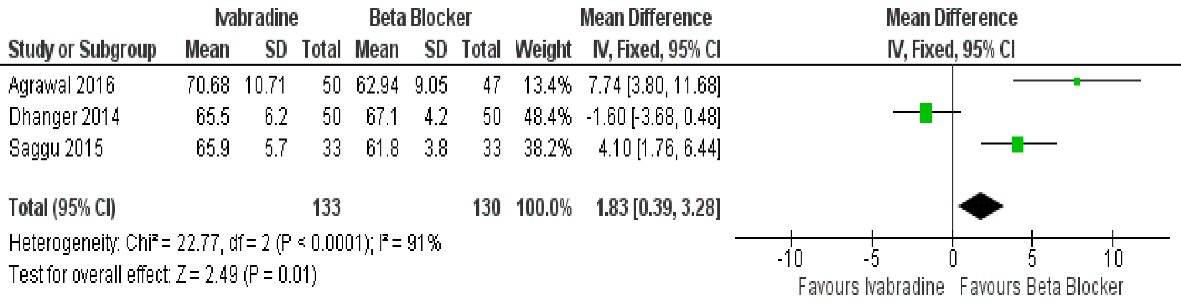 Click for large image | Figure 7. Forest plot showing the resting HR achieved with ivabradine versus beta-blocker in patients with MS in sinus rhythm. |
| Discussion | ▴Top |
Pooled analysis from five identified trials showed that among patients with MS in sinus rhythm, ivabradine was better compared to beta-blockers in total exercise duration, maximum HR achieved after exercise, and work capacity; inferior to beta-blockers in resting HR achieved; and comparable to beta-blockers in terms of mean gradient.
The demonstration of the better efficacy of ivabradine in improving exercise duration, maximum HR achieved after exercise, and improvement in work capacity, as compared to beta-blocker, is a new and significant finding. This is in contrast to the earlier meta-analysis by Adviento et al [20], where ivabradine was not significantly different to beta-blocker with regards to these clinical parameters. Individually, the included studies [17-19] showed that ivabradine and beta-blockers were able to similarly improve these outcomes, but in our study, it appears that ivabradine is in fact better. Dhanger [21] was able to show a statistically significant improvement in exercise duration from baseline among symptomatic MS patients in sinus rhythm, when treated with ivabradine for 3 months. Further, when compared head-to-head with atenolol, it was also statistically significant. This likely pulled the point estimate to favor ivabradine. Intuitively, we would expect that with an increase in time of exercise duration, work capacity should also improve, as these are related clinical outcomes. True enough, we are able to demonstrate this.
Exercise duration and work capacity are most likely related to the maximum HR that is achieved. Maximum HR achieved during or after exertion or exercise is an important clinical parameter for patients with MS, whether in sinus rhythm or not. As mentioned, the transmitral pressure gradient is dependent on HR, with a quadrupling of transvalvular pressure for a doubling of the HR [5]. With increase in HR with exercise, the transvalvular and pulmonary capillary wedge pressures (PCWP) also increase, resulting to dyspnea. This principle underlies the use of HR controllers, such as beta- and calcium channel blockers, in MS, as indicated in the guidelines [13]. HR control improves diastolic filling and prevents the rise of PCWP [18]. Several studies have demonstrated the value of beta-blockers in the hemodynamics and symptom improvement of MS [7-10]. In our study, we have shown that ivabradine was able to achieve a significantly lower maximal HR after exercise, compared to guideline-based HR controllers. This makes ivabradine a welcome alternative to beta-blockers, especially among patients with contraindications to beta-blocker therapy. The relative safety of ivabradine compared to beta-blockers is well known [11].
The observed favorable trend for ivabradine is likely related to the fact that the effect of ivabradine is more marked during higher heart rates i.e. as in exercise, as the If, or funny channel, can only be blocked when it is open, and it is open more frequently at higher heart rates; whereas the HR controlling effect of beta-blocker decreases during exertion [16]. This concept can also explain the observed superiority of beta-blocker over ivabradine in resting HR.
Mean transvalvular gradient was reduced by both ivabradine and beta-blocker, with the trend favoring ivabradine. This was, however, not statistically significant. As already noted, mean gradient is largely affected by HR, and we would expect a significant reduction in mean gradient with ivabradine, especially with exercise. Other factors may contribute to this apparent attenuation on the effect of ivabradine. The severity of the mitral valve stenosis may play a role. For example, Dhanger [21] started at a baseline mean gradient of about 12 mm Hg, while Saggu [17] started at about 10 mm Hg. In both studies, ivabradine and beta-blocker both significantly improved mean gradient, but this slight difference in the hemodynamic severity of the lesions may have spelled the difference to achieving statistical significance between them.
The results appear to favorably support the use of ivabradine as a viable option for patients with MS in sinus rhythm, especially to those who are intolerant to beta-blockers, or to whom beta-blockers are contraindicated [22]. Ergun et al [23], however, issued a cautionary letter to using ivabradine in MS in sinus rhythm patients, as ironically, ivabradine is associated with a 15% increase in incidence in atrial fibrillation, as shown in a meta-analysis [24]. A recent and larger meta-analysis involving about 40,000 patients corroborated this observation, showing a significantly higher incidence of atrial fibrillation with ivabradine compared to controls, with a 24% relative risk increase noted [25].
The standard for treatment in symptomatic and/or severe MS would still be surgery or valvuloplasty [6]. Guideline-based medical therapy would include heart failure management, and rate control for those who are symptomatic with exertion-induced tachycardia and dyspnea, and those with mild to moderate lesions [16, 18, 20].
Limitations
The search strategy results indicate that there are only a few studies that have examined the value of ivabradine in MS, and we are primarily limited by the small trials that are available. Interest, however, is growing in recent years, especially in the developing world where rheumatic MS remains a major health concern. We are also limited by the mostly open-label design of the trials, which is a major source of bias. We also did not analyze data according to dose. Available studies mostly have unclear risks of bias, and duration of the studies is short, especially when considering safety outcomes, such as the incidence of atrial fibrillation. Better-designed and longer trials would shed further light on these questions.
A strength of this meta-analysis is that we were able to keep heterogeneity to a minimum level in most of the outcomes that we measured, and we were able to avoid publication bias, as shown in our funnel plot analysis. We also demonstrated clinically important outcomes such as exercise duration and work capacity, and related these to hemodynamic outcomes such as HR and mean gradient.
Conclusions
Ivabradine is better or comparable to beta-blockers in terms of the outcomes measured, and may be considered as an alternative for patients with MS in sinus rhythm who are intolerant to beta-blockers.
| References | ▴Top |
- Seckeler MD, Hoke TR. The worldwide epidemiology of acute rheumatic fever and rheumatic heart disease. Clin Epidemiol. 2011;3:67-84.
doi pubmed - Carapetis JR, Steer AC, Mulholland EK, Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis. 2005;5(11):685-694.
doi - Mayosi Bongani. Natural history, screening, and management of rheumatic heart disease. http://www.uptodate.com/contents/natural-history-screening-and-management-of-rheumatic-heart-disease.
- Jara RD, Tumabiene KD. Valvuloplasty for rheumatic mitral stenosis: the Philippine experience. Phil Heart Center J. Jan - June 2013.
- Otto CM, Bonow RO. Valvular Heart Disease. In: Mann DL, Zipes DP, Libby P, Bonnow RO, Braunwald E. Braunwald's heart disease: a textbook of cardiovascular medicine. 10th edition. USA: Saunders, 2015.
- Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, 3rd, Guyton RA, O'Gara PT, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63(22):2438-2488.
doi pubmed - Stoll BC, Ashcom TL, Johns JP, Johnson JE, Rubal BJ. Effects of atenolol on rest and exercise hemodynamics in patients with mitral stenosis. Am J Cardiol. 1995;75(7):482-484.
doi - Kumar R, Saran RK, Dwivedi SK, Narain VS, Puri VK, Hasan M, Chandra N, et al. Beneficial effects of long-term metoprolol therapy on cardiac haemodynamics in patients with mitral stenosis in sinus rhythm - a randomised clinical trial. Indian Heart J. 1994;46(6):297-301.
pubmed - Monmeneu Menadas JV, Marin Ortuno F, Reyes Gomis F, Jordan Torrent A, Garcia Martinez M, Bodi Peris V, Garcia de Burgos de Rico E. Beta-blockade and exercise capacity in patients with mitral stenosis in sinus rhythm. J Heart Valve Dis. 2002;11(2):199-203.
pubmed - Sulit DV, Punzalan FE. Beta-blockers for mitral stenosis. Cochrane Database of Systematic Reviews 2001, Issue 4. Art. No.: CD003337.
doi - Sulfi S, Timmis AD. Ivabradine — the first selective sinus node I(f) channel inhibitor in the treatment of stable angina. Int J Clin Pract. 2006;60(2):222-228.
doi pubmed - Task Force Members, Montalescot G, Sechtem U, Achenbach S, Andreotti F, Arden C, Budaj A, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34(38):2949-3003.
doi pubmed - Yancy CW. et al. 2016 ACC/AHA/HFSA focused update on new pharmacological therapy for heart failure: an update of the 2013 ACCF/AHA guideline for the management of heart failure a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Card Fail. 2016.
- Swedberg K, Komajda M, Bohm M, Borer JS, Ford I, Dubost-Brama A, Lerebours G, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet. 2010;376(9744):875-885.
doi - Fox K, Ford I, Steg PG, Tendera M, Ferrari R, Investigators B. Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372(9641):807-816.
doi - Agrawal V, Kumar N, Lohiya B, Sihag BK, Prajapati R, Singh TB, Subramanian G. Metoprolol vs ivabradine in patients with mitral stenosis in sinus rhythm. Int J Cardiol. 2016;221:562-566.
doi pubmed - Saggu DK, Narain VS, Dwivedi SK, Sethi R, Chandra S, Puri A, Saran RK. Effect of ivabradine on heart rate and duration of exercise in patients with mild-to-moderate mitral stenosis: a randomized comparison with metoprolol. J Cardiovasc Pharmacol. 2015;65(6):552-554.
doi pubmed - Parakh N, Chaturvedi V, Kurian S, Tyagi S. Effect of ivabradine vs atenolol on heart rate and effort tolerance in patients with mild to moderate mitral stenosis and normal sinus rhythm. J Card Fail. 2012;18(4):282-288.
doi pubmed - Rajesh GN, Sajeer K, Sajeev CG, Bastian C, Vinayakumar D, Muneer K, Haridasan V, et al. A comparative study of ivabradine and atenolol in patients with moderate mitral stenosis in sinus rhythm. Indian Heart J. 2016;68(3):311-315.
doi pubmed - Adviento JB, Chiong LL, Aherrera JA, and Ramos JD. Ivabradine versus beta blockers in mitral stenosis in sinus rhythm: a meta-analysis of randomized controlled trials (Unpublished).
- Dhanger MK. Comparative study of ivabradine versus atenolol in symptomatic mitral stenosis patients. Indian Heart Journal. 2014;66:S133.
doi - Feldman AM. Ivabradine in Cardiovascular Disease: Heart Rate Isn't Everything. J Cardiovasc Pharmacol. 2015;65(6):549-551.
doi pubmed - Ergun G, Dogan V, Basaran O, Biteker M. Metoprolol vs ivabradine in patients with mitral stenosis. Int J Cardiol. 2016;223:108.
doi pubmed - Martin RI, Pogoryelova O, Koref MS, Bourke JP, Teare MD, Keavney BD. Atrial fibrillation associated with ivabradine treatment: meta-analysis of randomised controlled trials. Heart. 2014;100(19):1506-1510.
doi pubmed - Tanboga IH, Topcu S, Aksakal E, Gulcu O, Aksakal E, Aksu U, Oduncu V, et al. The risk of atrial fibrillation with ivabradine treatment: a meta-analysis with trial sequential analysis of more than 40000 patients. Clin Cardiol. 2016;39(10):615-620.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


